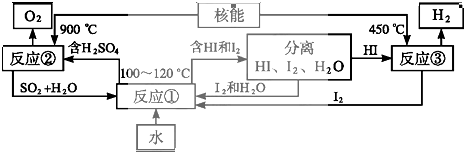
��Ŀ����
16��ijͬѧ�����в�������100mL 1.00mol/LNaCl��Һ���ټ�������NaCl�����������
�ڳ���NaCl���壻
�۽��ƺõ�NaCl��������ձ��У�����������ˮ�ܽ⣻
�ܽ��ձ��е���Һת������ƿ��������������ˮϴ���ձ��ڱ�2��3�Σ�ϴ��ҺҲע������ƿ��
��������ƿ�м�����ˮ���̶��ߣ�
��ش��й����⣺
��1����������NaCl���������5.85 g����������ƽʵ�ʳƵ�5.9 g��
��2��Ϊ�˼����ܽ⣬���Բ�ȡ�Ĵ�ʩ�ǽ��裮
��3��ʹ������ƿ�Ĺ����100 mL��
��4��������ʵ������У�����������;�ǽ��衢������
��5��������ձ��е���Һת�Ƶ�����ƿʱ������������ƿ�⣬�����ɵ���Һ�����ʵ�ʵ��Ũ�ȱ���Ҫ��С�����С�����ˣ�
��6���ý�ͷ�ι�������ƿ�м�ˮʱ����С��Һ�泬���˿̶ȣ���ȷ�����ķ�����D������ţ���
A����������Һ�壬ʹ��Һ����̶�������
B��С�ļ�������ƿ����������ʹ��Һ����̶�������
C�����������һ������NaCl
D����������
��7�������ͬѧ��5.00mol/LNaCl��Һ���ƣ�����Ҫ5.00mol/LNaCl��Һ20 mL��
���� ��1������m=CVM�����Ȼ��Ƶ�������������ƽֻ�ܳ�����0.1g��
��2�������ܼ��ٹ�����ܽ⣻
��3��ѡȡ������ƿ���Ӧ�õ��ڻ��Դ���������Һ�������
��4����ʵ������У����ܽ����Һ�IJ������õ���������
��5������c=$\frac{n}{V}$��������ʵ����ʵ���n����Һ�����V�ı仯��������������
��6������ʵ��ԭ�����������Ʋ�����
��7��������Һϡ�Ͷ���CŨVŨ=CϡVϡ�����㣻
��� �⣺��1��������Ȼ��Ƶ�����m=CVM=1.00mol/L��0.1L��58.5g/mol=5.85g��������������ƽֻ�ܳ�����0.1g����Ӧ������5.9g�Ȼ��ƣ��ʴ�Ϊ��5.85��5.9g��
��2��Ϊ�˼��ٹ�����ܽ���Բ��ý���ķ������ʴ�Ϊ�����裻
��3��ʵ������100mL������ƿ������Ӧ��ʹ��100mL������ƿ���ʴ�Ϊ��100��
��4����ʵ������У����ܽ����Һ�IJ������õ������������ܽ�ʱ���������������ǽ��裬����Һʱ���������������������ʴ�Ϊ�����裬������
��5�����ձ��е���Һת�Ƶ�����ƿʱ������������ƿ�⣬��ᵼ�����ʵ���ʧ����ҺŨ��ƫС���ʴ�Ϊ��С��
��6���ý�ͷ�ι�������ƿ�м�ˮʱ����С��Һ�泬���˿̶ȣ����ֲ���Ӧ�������ƣ���ѡD��
��7���������5mol/L���Ȼ�����Һ�����ΪVmL��������Һϡ�Ͷ���CŨVŨ=CϡVϡ��֪��5mol/L��VmL=1mol/L��100mL
���V=20mL���ʴ�Ϊ��20mL��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƹ����еļ���������������ڻ�������Ŀ���ѶȲ���

 �ظ���ʦ�㲦ϵ�д�
�ظ���ʦ�㲦ϵ�д�| A�� | ���ַ�ɢϵ�з�ɢ����������� | |
| B�� | �������ɲ����գ��õ��IJ��ﶼ������������ | |
| C�� | FeCl2��FeCl3����Һ���ȶ����ڣ���Fe��OH��3���岻���ȶ����� | |
| D�� | һ�������¿����ת����FeCl2��Һ��FeCl3��Һ��Fe��OH��3���� |
| A�� | þ���Ͻ��ܶȽ�С��ǿ�ȸߣ����������캽�����IJ��� | |
| B�� | ���Է����еĻ�ѧ��Ӧ�������ǡ�H��0����S��0 | |
| C�� | CaC2�Ż�ʱ��ʹ����ĭ�������� | |
| D�� | ��ͭ��ͭп�Ͻ�������ͭ��ײ���ͭ�� |
�����ڿ�����ȼ�� �ں���Ļ������ڿ��������� ���������ƺ����ᷴӦ �������ڿ�������ȫȼ�� ��ͭ����Ũ���ᷴӦ��
| A�� | ֻ�Т٢ۢ� | B�� | ֻ�Т٢� | C�� | ֻ�Т٢ۢܢ� | D�� | �٢ڢۢܢ� |
| A�� | ��ϩ�Ľṹ��ʽΪCH2CH2 | B�� | CH4���ӵ����ģ�ͣ� | ||
| C�� | ȩ���ĵ���ʽ��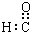 | D�� | �����ǵ����ʽ��ʵ��ʽ����CH2O |
��Al$\frac{ϡ����}{\;}$AlCl3$\stackrel{NaOH��Һ}{��}$Al��OH��3��
��Al$\stackrel{NaOH��Һ}{��}$Na[Al��OH��4]$\frac{ϡ����}{\;}$Al��OH��3
��
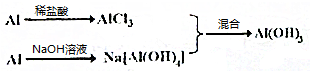
| ���� | ����1mol Al��OH��3�����Լ������ʵ��� | |
| n HCl/mol | n NaOH/mol | |
| �� | ||
| �� | ||
| �� | ||