��Ŀ����
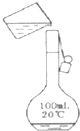
ʵ�����ɷ�ͭм�Ƶ�����CuSO4?5H2O����ʵ��������ͼ��

��1��ϴ��ͭм���۵��Լ�Ϊ5%Na2CO3��Һ������������Na2CO3��Һ��______�ԣ�
��2���ܽ�ͭмһ�ַ����ǣ���ͭм���뵽ϡ������˫��ˮ�Ļ��Һ�в���30��40��ˮԡ���ȣ�һ��ʱ���ͭ��ȫ�ܽ⣬�õ�����ͭ��Һ��
�ٸ÷�Ӧ�Ļ�ѧ����ʽΪ______��
�ڷ�Ӧ�¶Ȳ��ܳ���40���ԭ����______��
��3��������ͭ��Һ��õ����IJ�������Ϊ______��______�����ˡ�ϴ�ӡ����

��1��ϴ��ͭм���۵��Լ�Ϊ5%Na2CO3��Һ������������Na2CO3��Һ��______�ԣ�
��2���ܽ�ͭмһ�ַ����ǣ���ͭм���뵽ϡ������˫��ˮ�Ļ��Һ�в���30��40��ˮԡ���ȣ�һ��ʱ���ͭ��ȫ�ܽ⣬�õ�����ͭ��Һ��
�ٸ÷�Ӧ�Ļ�ѧ����ʽΪ______��
�ڷ�Ӧ�¶Ȳ��ܳ���40���ԭ����______��
��3��������ͭ��Һ��õ����IJ�������Ϊ______��______�����ˡ�ϴ�ӡ����
��1��Na2CO3��Һ�д���ˮ�����ӷ�ӦΪCO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-����Һ�ʼ��ԣ������ڼ��������·���ˮ�⣬����ϴ��ͭм���۵��Լ�Ϊ5%Na2CO3��Һ������������Na2CO3��Һ�ļ��ԣ��ʴ�Ϊ���
��2����˫��ˮ���������ԣ�ͭм���뵽ϡ������˫��ˮ�Ļ��Һ�У�˫��ˮ��ͭ������Cu2+��Cu2+��SO42-��ϵõ���������ͭ����ӦΪCu+H2O2+H2SO4=CuSO4+2H2O���ʴ�Ϊ��Cu+H2O2+H2SO4=CuSO4+2H2O��
�ڹ��������ֽ⣬��Ӧ�¶Ȳ��ܳ���40�棬��ֹH2O2�ֽ⣬�ʴ�Ϊ����ֹH2O2�ֽ⣻
��3������ͭ���ܽ�����¶����߶������ýᾧ���ᴿ���ڴֲ�Ʒ����ͭ�У�������ˮ�����ȳɱ�����Һ�����ȹ��˳�ȥ���������ʣ�����Ũ������Һ��ȴ����������ͭ���õ���������ͭ���ʴ�Ϊ������Ũ������ȴ�ᾧ��
��2����˫��ˮ���������ԣ�ͭм���뵽ϡ������˫��ˮ�Ļ��Һ�У�˫��ˮ��ͭ������Cu2+��Cu2+��SO42-��ϵõ���������ͭ����ӦΪCu+H2O2+H2SO4=CuSO4+2H2O���ʴ�Ϊ��Cu+H2O2+H2SO4=CuSO4+2H2O��
�ڹ��������ֽ⣬��Ӧ�¶Ȳ��ܳ���40�棬��ֹH2O2�ֽ⣬�ʴ�Ϊ����ֹH2O2�ֽ⣻
��3������ͭ���ܽ�����¶����߶������ýᾧ���ᴿ���ڴֲ�Ʒ����ͭ�У�������ˮ�����ȳɱ�����Һ�����ȹ��˳�ȥ���������ʣ�����Ũ������Һ��ȴ����������ͭ���õ���������ͭ���ʴ�Ϊ������Ũ������ȴ�ᾧ��

��ϰ��ϵ�д�
�����Ŀ

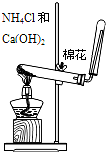
 ����K2CO3�е�K+
����K2CO3�е�K+