��Ŀ����
�ϳɰ����Ȼ�ѧ����ʽΪ��N2��g��+3H2��g��?2NH3��g������H=-92.4kJ?mol-1���ֽ�1mol N2��g����3mol H2��g������һ�ݻ�Ϊ2L���ܱ������У���500���½��з�Ӧ��10minʱ�ﵽƽ�⣬NH3���������Ϊ�أ�����˵������ȷ���ǣ�������
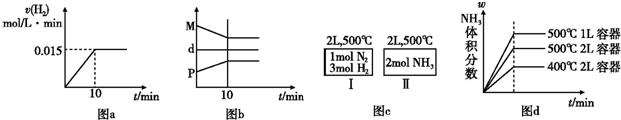

| A�����ﵽƽ��ʱ�������ϵ�ų�9.24 kJ��������H2��Ӧ���ʱ仯������ͼa��ʾ | B����Ӧ�����У��������ƽ����Է�������ΪM����������ܶ�Ϊd���������ѹǿΪP�����߹�ϵ��ͼb | C����ͼc��ʾ��������͢�ﵽƽ��ʱ��NH3���������Ϊ�أ���������ų���������������������֮��Ϊ92.4 kJ | D������ʼ��������Ϊ1 mol N2��3 mol H2���ڲ�ͬ�����´ﵽƽ��ʱ��NH3����������仯��ͼd��ʾ |
������A���ﵽƽ��ʱ�ų�������Ϊ9.24kJ�����Ȼ�ѧ����ʽ��֪�μӷ�Ӧ�����������ʵ���Ϊ0.3mol��10min��������ƽ������Ϊ0.015mol/��L?min������Ӧ����ӦΪ�ɸߵ��ͣ�
B�������������������䣬������������䣬���������ܶ�Ϊ��ֵ���淴Ӧ���С������������ʵ�����С����������ƽ����Է�����������������ѹǿ���ͣ�
C�����º����£��������а���ѧ������ת��ΪN2��H2���ɵ�N21mol��H23mol����������������Ϊ��Чƽ�⣮ƽ��ʱ�����ڶ�Ӧ���������ʵ�����ȣ���ƽ��ʱ������N2Ϊnmol������Ȼ�ѧ����ʽ���㣻
D������ѹǿƽ��������Ӧ�����ƶ��������¶�ƽ�����淴Ӧ�����ƶ����ݴ��ж�ƽ��ʱNH3�����������
B�������������������䣬������������䣬���������ܶ�Ϊ��ֵ���淴Ӧ���С������������ʵ�����С����������ƽ����Է�����������������ѹǿ���ͣ�
C�����º����£��������а���ѧ������ת��ΪN2��H2���ɵ�N21mol��H23mol����������������Ϊ��Чƽ�⣮ƽ��ʱ�����ڶ�Ӧ���������ʵ�����ȣ���ƽ��ʱ������N2Ϊnmol������Ȼ�ѧ����ʽ���㣻
D������ѹǿƽ��������Ӧ�����ƶ��������¶�ƽ�����淴Ӧ�����ƶ����ݴ��ж�ƽ��ʱNH3�����������
����⣺A����Ӧ��ʼ������ԽϿ죬��ƽ��ǰ��Ӧ������Խ��������Է�Ӧ����ӦΪ�ɸߵ��ͣ��ﵽƽ��ʱ�ų�������Ϊ9.24kJ�����Ȼ�ѧ����ʽ��֪�μӷ�Ӧ�����������ʵ���Ϊ0.3mol��10min��������ƽ������Ϊ0.015mol/��L?min������ͷ�Ӧ����ӦС��0.015mol/��L?min������A����
B�������������������䣬������������䣬���������ܶ�Ϊ��ֵ���淴Ӧ���С������������ʵ�����С����������ƽ����Է�����������������ѹǿ���ͣ���B����
C�����º����£��������а���ѧ������ת��ΪN2��H2���ɵ�N21mol��H23mol����������������Ϊ��Чƽ�⣮����;���дﵽƽ��ʱ��NH3�����������Ϊ�أ�ƽ��ʱ�����ڶ�Ӧ���������ʵ�����ȣ���ƽ��ʱ������N2Ϊnmol�����������зų�������Ϊ��1-n��mol��92.4kJ/mol=92.4��1-n��kJ�������������յ�����Ϊnmol��92.4kJ/mol=92.4nkJ���ݢ��зų���������������յ�����֮��Ϊ92.4��1-n��kJ+92.4nkJ=92.4kJ����C��ȷ��
D����ʼ��������Ͼ�Ϊ1molN2��3molH2���ڲ�ͬ�����´ﵽƽ��ʱ���Ƚ�500�桢2L��500�桢1L�����Сѹǿ��Ӧ���ʿ죬�ﵽƽ��ʱ��̣�ƽ��������Ӧ�ƶ���ƽ��ʱNH3�����������ԽϴȽ�500�桢2L��400�桢2L���¶�����Ӧ���ʿ죬�ﵽƽ��ʱ��̣�ƽ�����淴Ӧ�ƶ���ƽ��ʱNH3�����������Խ�С����D����
��ѡC��
B�������������������䣬������������䣬���������ܶ�Ϊ��ֵ���淴Ӧ���С������������ʵ�����С����������ƽ����Է�����������������ѹǿ���ͣ���B����
C�����º����£��������а���ѧ������ת��ΪN2��H2���ɵ�N21mol��H23mol����������������Ϊ��Чƽ�⣮����;���дﵽƽ��ʱ��NH3�����������Ϊ�أ�ƽ��ʱ�����ڶ�Ӧ���������ʵ�����ȣ���ƽ��ʱ������N2Ϊnmol�����������зų�������Ϊ��1-n��mol��92.4kJ/mol=92.4��1-n��kJ�������������յ�����Ϊnmol��92.4kJ/mol=92.4nkJ���ݢ��зų���������������յ�����֮��Ϊ92.4��1-n��kJ+92.4nkJ=92.4kJ����C��ȷ��
D����ʼ��������Ͼ�Ϊ1molN2��3molH2���ڲ�ͬ�����´ﵽƽ��ʱ���Ƚ�500�桢2L��500�桢1L�����Сѹǿ��Ӧ���ʿ죬�ﵽƽ��ʱ��̣�ƽ��������Ӧ�ƶ���ƽ��ʱNH3�����������ԽϴȽ�500�桢2L��400�桢2L���¶�����Ӧ���ʿ죬�ﵽƽ��ʱ��̣�ƽ�����淴Ӧ�ƶ���ƽ��ʱNH3�����������Խ�С����D����
��ѡC��
���������⿼��ƽ���ƶ�ͼ��Чƽ�⡢����ƽ�⼰��Ӧ���ʵ�Ӱ��ȣ��Ѷ��еȣ�ע��Dѡ���ж�һ������ȹ���ƽԭ������ã�

��ϰ��ϵ�д�
 һ����������ϵ�д�
һ����������ϵ�д�
�����Ŀ
 2NH3�ǹ�ҵ�Ϻϳɰ�����Ҫ��Ӧ��
2NH3�ǹ�ҵ�Ϻϳɰ�����Ҫ��Ӧ��
 2NH3��g����H=2��E1-E3����-2��E3-E1��kg/mol
2NH3��g����H=2��E1-E3����-2��E3-E1��kg/mol 2NH3��g��+92.2kJ/mol
2NH3��g��+92.2kJ/mol
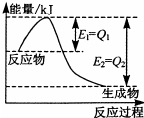 ���û�ѧ��Ӧԭ���о��������ȵȵ��ʼ��仯����ķ�Ӧ����Ҫ�����壮
���û�ѧ��Ӧԭ���о��������ȵȵ��ʼ��仯����ķ�Ӧ����Ҫ�����壮