��Ŀ����
A��B��C��D��E��F���ֶ�����Ԫ�أ�ԭ��������������A�ǵؿ��к�����ߵ�Ԫ�أ�C�ǵؿ��к�����ߵĽ���Ԫ�أ�A��Eͬ���塣DԪ��ԭ�������������Ǵ�����������һ�롣B��C��Ԫ��ԭ�ӵ�����������֮�͵���DԪ��ԭ�ӵ�������������C��D��Ԫ��ԭ������������֮�͵���FԪ��ԭ�ӵ�������������
�ش��������⣺
(1��Ԫ��A��Ԫ�����ڱ��е�λ���� ��
��2�� д��B��E��Ԫ���γɻ�����ĵ���ʽ�� ��������ѧ������ ��
��3��A��D��E��F����̬�⻯����ȶ���˳�� ���û�ѧʽ��ʾ��
��4��д��C��E��Ӧ������������ˮ�������Ӧ�Ļ�ѧ����ʽΪ��
��5��C��D����������ֱ���B���������ﷴӦ�����ӷ���ʽ��
��
�ش��������⣺
(1��Ԫ��A��Ԫ�����ڱ��е�λ���� ��
��2�� д��B��E��Ԫ���γɻ�����ĵ���ʽ�� ��������ѧ������ ��
��3��A��D��E��F����̬�⻯����ȶ���˳�� ���û�ѧʽ��ʾ��
��4��д��C��E��Ӧ������������ˮ�������Ӧ�Ļ�ѧ����ʽΪ��
��5��C��D����������ֱ���B���������ﷴӦ�����ӷ���ʽ��
��
(1)�ڶ����ڣ���A�� ��
(2)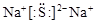 ���Ӽ�
���Ӽ�
(3) H2O> HCl>H2S>SiH4
(4)2Al(OH)3 +3 H2SO4 ="=" Al2(SO4)3 +6H2O
(5)Al2O3 +2OH�� ==2AlO2�� +H2O SiO2 + 2OH�� ==SiO32�� +H2O
(2)
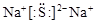 ���Ӽ�
���Ӽ� (3) H2O> HCl>H2S>SiH4
(4)2Al(OH)3 +3 H2SO4 ="=" Al2(SO4)3 +6H2O
(5)Al2O3 +2OH�� ==2AlO2�� +H2O SiO2 + 2OH�� ==SiO32�� +H2O
���������A�ǵؿ��к�����ߵ�Ԫ�أ���AΪO��C�ǵؿ��к�����ߵĽ���Ԫ�أ���CΪAl��A��Eͬ���壬��EΪS��DԪ��ԭ�������������Ǵ�����������һ��,��DΪSi��B��C��Ԫ��ԭ�ӵ�����������֮�͵���DԪ��ԭ�ӵ���������������BΪNa��C��D��Ԫ��ԭ������������֮�͵���FԪ��ԭ�ӵ���������������FΪCl������A��B��C��D��E��F�ֱ�ΪO��Na��Al��Si��S��Cl��(1��Ԫ��A��Ԫ�����ڱ��е�λ���ǣ��ڶ����ڣ���A�壻��2�� B��E��Ԫ���γɻ�����Ϊ���ƣ������ʽΪ
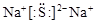 ��������ѧ������Ϊ���Ӽ�����3������Ԫ�صĵ縺��Խ����̬�⻯��Խ�ȶ�������A��D��E��F����̬�⻯����ȶ���˳��Ϊ��H2O> HCl>H2S>SiH4����4��C��E��Ӧ������������ˮ����ֱ�Ϊ�������������ᣬ���߷����Ļ�ѧ��Ӧ����ʽΪ��2Al(OH)3 +3 H2SO4 ="=" Al2(SO4)3 +6H2O��
��������ѧ������Ϊ���Ӽ�����3������Ԫ�صĵ縺��Խ����̬�⻯��Խ�ȶ�������A��D��E��F����̬�⻯����ȶ���˳��Ϊ��H2O> HCl>H2S>SiH4����4��C��E��Ӧ������������ˮ����ֱ�Ϊ�������������ᣬ���߷����Ļ�ѧ��Ӧ����ʽΪ��2Al(OH)3 +3 H2SO4 ="=" Al2(SO4)3 +6H2O����5��C��D��������ֱ�Ϊ�������Ͷ������裬�����������Ƶķ�Ӧ�����ӷ���ʽ�ֱ�Ϊ��
Al2O3 +2OH�� ==2AlO2�� +H2O SiO2 + 2OH�� ==SiO32�� +H2O��
���������⿼����Ԫ�����ڱ���Ԫ�������ɡ�Ԫ�ؼ��仯��������Ĺؼ������ƶϳ�A��B��C��D��E��F�ֱ�ΪO��Na��Al��Si��S��Cl��Ȼ���ٽ�𣬱����Ѷ��еȡ�

��ϰ��ϵ�д�
�����Ŀ
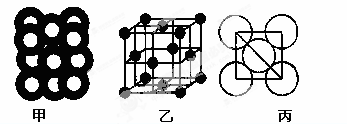
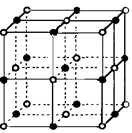
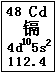
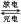 Cd(OH)2�� 2Ni(OH)2
Cd(OH)2�� 2Ni(OH)2 ��
�� 
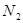 ��
�� 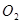
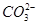 ��
�� 
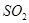 ��
��