��Ŀ����
��14�֣�
��A��B��C��D��E��F���ֶ�����Ԫ�أ���Ԫ��������Ϣ���±���
|
Ԫ�ر�� |
Ԫ��������Ϣ |
|
A |
A�ĵ������ܶ���С������ |
|
B |
B�ĵ���������ˮ���ҷ�Ӧ������ǿ������Һ�к������ֵ�������ͬ������������ |
|
C |
C��ԭ�����������������ڲ������������ |
|
D |
D��Bͬ���ڣ���������D�ļ����Ӱ뾶��С |
|
E |
B��C��E��ɵ�36���ӵĻ�����Y�Ǽ�������������Ҫ�ɷ� |
|
F |
FԪ���������������۵Ĵ�����Ϊ4 |
��1��д��������Y�����Ļ�ѧ������ ��
��2��D��E��F�ļ����Ӱ뾶�ɴ�С��˳���ǣ��û�ѧʽ��ʾ�� ��
��3��д�����־���A��B��C��F����Ԫ�صĻ���������Һ�����Ӧ�����ӷ���ʽ
��
��4����Fe��D������ɵĻ�����У���������F������������Ӧˮ�����ϡ��Һ������ȫ���ܽ⡣�����õ���Һ�м������������������Һ���������ij������˳�������ϴ�ӡ�������պ�õ�һ�ֹ��壬���������ָù����������ԭ����������ǡ����ȣ���ԭ�������D���ʵ���������Ϊ ��
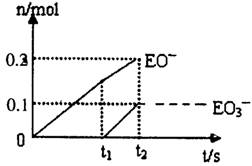
��5��һ������ʯ������ͨ��һ������E���ʡ�����ǡ����ȫ��Ӧ���������������ֺ�EԪ�ص����ӣ������������ӵ����ʵ�����n���뷴Ӧʱ�䣨t����������ͼ��ʾ����t2ʱ������������������Ϊ g����ʱ��Ӧ�Ļ�ѧ����ʽΪ ��
��6��A��B�γɵĻ�����BA���л��ϳ�����;�ܹ㷺�������Զ�ȡ�ܶ�����е����Ӷ�������Ӧ���ƻ����д�������Ҵ���Ӧ�Ļ�ѧ����ʽ ��
��1�����Ӽ������ۼ������Թ��ۼ�������1�֣�
��2��S2->Cl>Al3+
��3��H++HSO-3=SO2��+H2O
��4��30%
��5��37g��10Cl2+10Ca��OH��2=7CaCl2+2Ca��ClO��2+Ca��ClO3��2+10H2O
��6��NaH+CH3CH2OH=CH3CH2Ona+H2��
��������

 ��A��B��C��D��E���ֶ�����Ԫ�أ����ǵ�ԭ��������A��E����������֪A��Bԭ������ͬ�ĵ��Ӳ�������A��L���������K���������������C�ڿ�����ȼ��ʱ���ֻ�ɫ���棬C�ĵ����ڸ�������B�ĵ��ʳ�ַ�Ӧ�����Եõ���E������ɫ��ͬ�ĵ���ɫ��̬�����DΪԭ�Ӻ�����12�����ӵĶ��۽�������2.4��D�����ᷴӦʱ���ڱ�״���·ų�����2.24L��
��A��B��C��D��E���ֶ�����Ԫ�أ����ǵ�ԭ��������A��E����������֪A��Bԭ������ͬ�ĵ��Ӳ�������A��L���������K���������������C�ڿ�����ȼ��ʱ���ֻ�ɫ���棬C�ĵ����ڸ�������B�ĵ��ʳ�ַ�Ӧ�����Եõ���E������ɫ��ͬ�ĵ���ɫ��̬�����DΪԭ�Ӻ�����12�����ӵĶ��۽�������2.4��D�����ᷴӦʱ���ڱ�״���·ų�����2.24L��

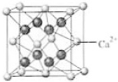 ��4��B��Ca�γɵľ���ľ�������ͼ��ʾ������Ca2+����λ����
��4��B��Ca�γɵľ���ľ�������ͼ��ʾ������Ca2+����λ����