��Ŀ����
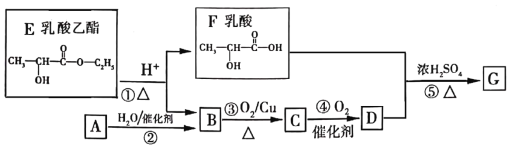
����Ŀ����֪��A������������������ܶ���15�����ܷ���������Ӧ��F�ķ���ʽΪC3H6O2���й����ʵ�ת����ϵ���£�

��ش�
��1��B�к��еĹ�����������__________����Ӧ�ķ�Ӧ����Ϊ__________��
��2��д��A����������Ӧ�Ļ�ѧ����ʽ__________��
��3��д����Ӧ�ܵĻ�ѧ����ʽ__________��
��4��д���л���F��NaOH��Һ��Ӧ�Ļ�ѧ����ʽ__________��
��5������˵����ȷ����__________��
A �л���D��ˮ��Һ�����ڱ걾�ķ���
B �л���B��C��E����������Ʒ�����Ӧ
C �л���F�л���E�����ñ���̼������Һ���з���
D �л���MΪ�߷��ӻ�����
���𰸡��ǻ� ����(��ȡ��)��Ӧ HCHO+4Ag(NH3)2OH ![]() 4Ag��+2H2O+(NH4)2CO3+6NH3�� 2CH3CH2OH+O2
4Ag��+2H2O+(NH4)2CO3+6NH3�� 2CH3CH2OH+O2![]() 2CH3CHO+2H2O CH3COOCH3+ NaOH
2CH3CHO+2H2O CH3COOCH3+ NaOH ![]() CH3COONa+CH3OH BC
CH3COONa+CH3OH BC
��������
��A������������������ܶ���15������֪��A����Է�������Ϊ30���ܷ���������Ӧ˵������-CHO����AΪ��ȩ��HCHO������ȩ�����������ӳɷ�Ӧ����CΪCH3OH���������ھƻ�ø�����������Ҵ������Ƴ�MΪ�����ǣ�BΪ�Ҵ����Ҵ���������������DΪ��ȩ����ȩ��������EΪ���ᣬ����ͼ״�����������Ӧ����FΪ����������ݴ˽��
��1���������Ϸ�����BΪ�Ҵ����ṹ��ʽΪC2H5OH�����еĹ������������ǻ�����Ӧ��������ͼ״�������Ӧ���������������Ӧ����Ϊ������Ӧ��Ҳ��ȡ����Ӧ��
�ʴ�Ϊ���ǻ�������(��ȡ��)��Ӧ��
��2��AΪHCHO������������Ӧ�Ļ�ѧ����ʽHCHO+4Ag(NH3)2OH ![]() 4Ag��+2H2O+(NH4)2CO3+6NH3����
4Ag��+2H2O+(NH4)2CO3+6NH3����
�ʴ�Ϊ��HCHO+4Ag(NH3)2OH ![]() 4Ag��+2H2O+(NH4)2CO3+6NH3����
4Ag��+2H2O+(NH4)2CO3+6NH3����
��3����Ӧ��Ϊ�Ҵ�����������������ȩ���仯ѧ����ʽΪ2CH3CH2OH+O2![]() 2CH3CHO+2H2O��
2CH3CHO+2H2O��
�ʴ�Ϊ��2CH3CH2OH+O2![]() 2CH3CHO+2H2O��
2CH3CHO+2H2O��
��4��FΪ����������ڼ���������ˮ�����������ƺͼ״�����Ӧ�Ļ�ѧ����ʽΪCH3COOCH3+ NaOH ![]() CH3COONa+CH3OH��
CH3COONa+CH3OH��
�ʴ�Ϊ��CH3COOCH3+ NaOH ![]() CH3COONa+CH3OH��
CH3COONa+CH3OH��
��5��A .�л���DΪ��ȩ������ȩ35%~40%��ˮ��Һ�׳Ƹ������֣������ڱ걾�ķ�������A����
B .�л���B��C��E�ֱ�Ϊ�Ҵ����״������ᣬ�����к����ǻ����Ȼ�������������Ʒ�����Ӧ����B��ȷ��
C. ��������л������ᣬ���뱥��̼������Һ�������ܽ⣬����������ܣ���Һ�ֲ㣬��ͨ����Һ���룬��C��ȷ��
D.�л���MΪ�����ǣ�Ϊ���ǣ����Ǹ߷��ӻ������D����
�ʴ�ѡ��BC��

����Ŀ���������״���;����㷺��Խ��Խ�����̼ҵĹ�ע����ҵ�ϼ״��ĺϳ�;�����ֶ���������ʵ������ģ��״��ϳɷ�Ӧ,��2 L�ܱ������ڣ�400 ��ʱ������ӦCO(g)+2H2(g) ![]() CH3OH(g)����ϵ��n(CO)��ʱ��ı仯�����
CH3OH(g)����ϵ��n(CO)��ʱ��ı仯�����

ʱ��(s) | 0 | 1 | 2 | 3 | 5 |
n(CO)(mol) | 0.020 | 0.011 | 0.008 | 0.007 | 0.007 |
��1��ͼ�б�ʾCH3OH �ı仯��������_______��
��2�����д�ʩ������߷�Ӧ���ʵ���_________(������Ӧ��ĸ���)��
a �����¶� b ������� c ����ѹǿ d ��ʱ�����CH3OH
��3������������˵����Ӧ�ﵽƽ��״̬����__________(������Ӧ��ĸ���)��
a.CO��H2��Ũ�ȱ��ֲ���
b.v(H2)=2 v(CO)
c.CO�����ʵ����������ֲ���
d.�����������ܶȱ��ֲ���
e.ÿ����1molCH3OH��ͬʱ��2molH-H������
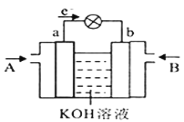
��4��CH3OH��O2�ķ�Ӧ�ɽ���ѧ��ת��Ϊ���ܣ��乤��ԭ����ͼ��ʾ��ͼ��CH3OH��__________����A��B��ͨ�룬b���ĵ缫��Ӧʽ��__________��