��Ŀ����
��֪25��ʱ����������ʵĵ���ƽ�ⳣ���������±���
| ��ѧʽ | CH3COOH | H2CO3 | HClO |
| ƽ�ⳣ�� | Ka=1.8��10-5 | Ka1=4.3��10-7 Ka2=5.6��10-11 | Ka=3.0��10-8 |
�ش��������⣺
��1�����ʵ���Ũ�Ⱦ�Ϊ0.1mol•L-1��������Һ��
A��CH3COOH B��Na2CO3 C��NaClO D��NaHCO3��
pH��С���������˳���� ;adcb������ĸ��ʾ��
��2�������£�0.1mol•L-1��CH3COOH��Һ��ˮϡ�����У����б���ʽ�����ݱ�����( )
A��c��H+�� B��c��H+��/c��CH3COOH��
C��c��H+��•c![]() ��OH-�� D��c��OH-��/c��H+��
��OH-�� D��c��OH-��/c��H+��
E��c��H+��•c( CH3COO-)/��CH3COOH��
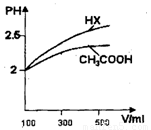
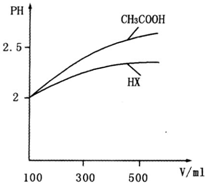
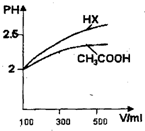
��3�������Ϊ100mL pH=2��CH3COOH��һԪ��HX����ˮϡ������pH����Һ����Ĺ�ϵ��ͼ��ʾ����HX�ĵ���ƽ�ⳣ�� ����CH3COOH�ĵ���ƽ�ⳣ��������ڡ�С�ڻ���ڣ��������� .
��4��25��ʱ��CH3COOH��CH3COONa�Ļ����Һ�������pH=6������Һ��
c��CH3COO-����c��Na+��= ���ȷֵ����c��CH3COO-��/c��CH3COOH��= ��
(1)ADCB (2)BD
(3)���ڣ�ϡ����ͬ������һԪ��HX��pH�仯�ȴ��������ǿ������ƽ�ⳣ����
(4)9.9��10-7 18
��������
�����������1������Խ��Խˮ�����ĵ���ƽ�ⳣ����֪HCO3-![]() �ĵ��볣����С����Na2CO3�ļ�����ǿ����ʵ��NaClO��NaHCO3�������CH3COOH����pH��С�����˳����ADCB��
�ĵ��볣����С����Na2CO3�ļ�����ǿ����ʵ��NaClO��NaHCO3�������CH3COOH����pH��С�����˳����ADCB��
��2��ϡ������c(H+)��С��K=![]() ��ϡ�����в��䣬c(CH3COO-)��С������
��ϡ�����в��䣬c(CH3COO-)��С������![]() ����c(H+)•c(OH-)���䣬
����c(H+)•c(OH-)���䣬![]() ���������������BD��
���������������BD��
��3������ͼ���֪HXϡ�ͺ�pH�仯������HX������ǿ��CH3COOH��HX�ĵ���ƽ�ⳣ������CH3COOH�ĵ���ƽ�ⳣ����
��4�����ݵ���غ�c![]() (H+)+c(Na+)=c(OH-)+c(CH3COO-)����c(CH3COO-)-c(Na+)=c(H+)-c(OH-)����Һ��pH=6����c(H+)=10-6mol/L��c(OH-)=10-8mol/L������c(CH3COO-)-c(Na+)=9.9��10-7��K=
(H+)+c(Na+)=c(OH-)+c(CH3COO-)����c(CH3COO-)-c(Na+)=c(H+)-c(OH-)����Һ��pH=6����c(H+)=10-6mol/L��c(OH-)=10-8mol/L������c(CH3COO-)-c(Na+)=9.9��10-7��K=![]() ����
����![]() =
=![]() =18��
=18��
���㣺����ƽ��
�����������ۺ��Խ�ǿ���ѶȽϴ���ҪҪ���ú�ƽ�ⳣ����

��֪25��ʱ����������ʵĵ���ƽ�ⳣ�����������ʾ��
| ��ѧʽ | CH3COOH | H2CO3 | HClO | |
| ����ƽ�ⳣ�� | Ka��1.8��10��5 | Kal��4.3��10��7 | Ka2��5.6��10��11 | Ka��3.0��10��8 |
�ش��������⣺
(1)���ʵ���Ũ�Ⱦ�Ϊ0.1mol��L��1��������Һ��
a��CH3COONa b��Na2CO3 c��NaClO d��NaHCO3
pH��С�������е�˳����_______________(�ñ����д)��
(2)�����£�0.1mol��L��1CH3COOH��Һ��ˮϡ�����У����б���ʽ�����ݱ����� ��
A��c(H��) B��c(H��)/c(CH3COOH) C��c(H��)��c(OH��)
D��c(OH��)/c(H��) E��c(H��)��c(CH3COO��)/c(CH3COOH)
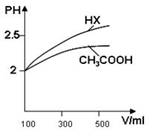
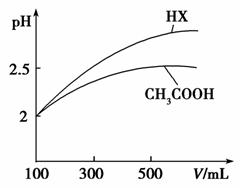
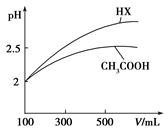
(3)�����Ϊ100mL pH��2��CH3COOH��һԪ��HX����ˮϡ������pH����Һ����Ĺ�ϵ��ͼ��ʾ����HX�ĵ���ƽ�ⳣ��_____(����ڡ�����С�ڡ����ڡ�)CH3COOH�ĵ���ƽ�ⳣ����������_____________________��
(4)25��ʱ��CH3COOH��CH3COONa�Ļ����Һ�������pH��6������Һ��
c(CH3COO��)��c(Na��)�� mol��L��1(�ȷֵ)��
(5)��״���£���1.12L CO2ͨ��100mL 1mol��L��1��NaOH��Һ�У�����Һ������Ũ�ȷ���������е�ʽ��
��c (OH��)��2c(H2CO3)�� ��
��c(H��)��c(Na��)�� ��
��֪25��ʱ����������ʵĵ���ƽ�ⳣ�����������ʾ��
|
��ѧʽ |
CH3COOH |
H2CO3 |
HClO |
|
|
����ƽ�ⳣ�� |
Ka��1.8��10��5 |
Kal��4.3��10��7 |
Ka2��5.6��10��11 |
Ka��3.0��10��8 |
�ش��������⣺
(1)���ʵ���Ũ�Ⱦ�Ϊ0.1mol��L��1��������Һ��
a��CH3COONa b��Na2CO3 c��NaClO d��NaHCO3
pH��С�������е�˳����_______________(�ñ����д)��
(2)�����£�0.1mol��L��1CH3COOH��Һ��ˮϡ�����У����б���ʽ�����ݱ����� ��
A��c(H��) B��c(H��)/c(CH3COOH) C��c(H��)��c(OH��)
D��c(OH��)/c(H��) E��c(H��)��c(CH3COO��)/c(CH3COOH)
(3)�����Ϊ100mL pH��2��CH3COOH��һԪ��HX����ˮϡ������pH����Һ����Ĺ�ϵ��ͼ��ʾ����HX�ĵ���ƽ�ⳣ��_____(����ڡ�����С�ڡ����ڡ�)CH3COOH�ĵ���ƽ�ⳣ����������_____________________��
(4)25��ʱ��CH3COOH��CH3COONa�Ļ����Һ�������pH��6������Һ��
c(CH3COO��)��c(Na��)�� mol��L��1(�ȷֵ)��
(5)��״���£���1.12L CO2ͨ��100mL 1mol��L��1��NaOH��Һ�У�����Һ������Ũ�ȷ���������е�ʽ��
��c (OH��)��2c(H2CO3)�� ��
��c(H��)��c(Na��)�� ��
 ��֪25��ʱ����������ʵĵ���ƽ�ⳣ���������±���
��֪25��ʱ����������ʵĵ���ƽ�ⳣ���������±���