��Ŀ����
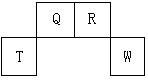
��14�֣��±�ΪԪ�����ڱ���һ���֣������Ԫ�آ٣����ڱ��е�λ�ã��û�ѧ����ش��������⣺
��1����д���ڵ�Ԫ�ط���
��2����д���۵����������ĵ���ʽ ��
��3���ȽϢݡ��ޡ����ԭ�Ӱ뾶�ɴ�С��˳��Ϊ(��Ԫ�ط��ű�ʾ)
��4�� �ȽϢۡ��ܡ������ۺ������������ǿ������˳����(����Ļ�ѧʽ��ʾ) ��
�ȽϢۡ��ܡ������ۺ������������ǿ������˳����(����Ļ�ѧʽ��ʾ) ��
��5��д����Ԫ�آ٣���������������Ӧˮ�������ǿ������ǿ������֮��Ļ�ѧ��Ӧ����ʽ
��6���ߢ���Ԫ����Ƚϣ������Խ�ǿ���� �������ƣ���������֤�ý��۵�ʵ���� �����ţ���
��a�����ڿ����з����Ѿõ�������Ԫ�صĿ�״���ʷֱ������ˮ��
��b������״����С��ͬ��������Ԫ�صĵ��ʷֱ��ͬŨ�ȵ����ᷴӦ
��c������״����С��ͬ������Ԫ�صĵ��ʷֱ����ˮ���ã��������̪��Һ
��d���Ƚ�������Ԫ�ص���̬�⻯����ȶ���
�� ���� ���� | IA | | 0 | |||||
| 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
| 2 | | �� | | �� | �� | | �� | |
| 3 | �� | �� | �� | �� | | | �� | |
��2����д���۵����������ĵ���ʽ ��
��3���ȽϢݡ��ޡ����ԭ�Ӱ뾶�ɴ�С��˳��Ϊ(��Ԫ�ط��ű�ʾ)
��4��
 �ȽϢۡ��ܡ������ۺ������������ǿ������˳����(����Ļ�ѧʽ��ʾ) ��
�ȽϢۡ��ܡ������ۺ������������ǿ������˳����(����Ļ�ѧʽ��ʾ) ����5��д����Ԫ�آ٣���������������Ӧˮ�������ǿ������ǿ������֮��Ļ�ѧ��Ӧ����ʽ
��6���ߢ���Ԫ����Ƚϣ������Խ�ǿ���� �������ƣ���������֤�ý��۵�ʵ���� �����ţ���
��a�����ڿ����з����Ѿõ�������Ԫ�صĿ�״���ʷֱ������ˮ��
��b������״����С��ͬ��������Ԫ�صĵ��ʷֱ��ͬŨ�ȵ����ᷴӦ
��c������״����С��ͬ������Ԫ�صĵ��ʷֱ����ˮ���ã��������̪��Һ
��d���Ƚ�������Ԫ�ص���̬�⻯����ȶ���
��1��Be ��2��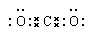 ��3�� Na ��Al �� F
��3�� Na ��Al �� F
��4�� HNO3��H2CO3 ��H2SiO3 ( ��H4SiO4) ��
HNO3��H2CO3 ��H2SiO3 ( ��H4SiO4) ��
��5��NaOH + HClO4 ="=" NaClO4 + H2O ��6�� þ ��b.c
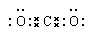 ��3�� Na ��Al �� F
��3�� Na ��Al �� F ��4��
 HNO3��H2CO3 ��H2SiO3 ( ��H4SiO4) ��
HNO3��H2CO3 ��H2SiO3 ( ��H4SiO4) ����5��NaOH + HClO4 ="=" NaClO4 + H2O ��6�� þ ��b.c
����Ԫ�����ڱ��Ľṹ��Ԫ�������ɵ�Ӧ�á�����Ԫ�������ڱ��е�λ�ÿ��жϢ١���ֱ���H��Be��C��N��F��Na��Mg��Al��Si��Cl��
��1���ԣ�2��̼�������������CO2�����ڹ��ۻ��������ʽΪ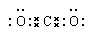 ��
��
��3��ͬ����Ԫ����������ԭ�Ӱ뾶��С��ͬ����Ԫ�����϶���ԭ�Ӱ뾶���������Ԣݡ��ޡ����ԭ�Ӱ뾶��С˳��ΪNa ��Al �� F��
��4���ǽ�����Խǿ����Ӧ����������ˮ��������Ծ�Խǿ���ǽ�������N��C��Si������������HNO3��H2CO3 ��H2SiO3��
��5��������Ԫ��������õĽ������ƣ��ǽ������ȣ����Է���ʽΪNaOH + HClO4 ="=" NaClO4 + H2O��
��6��ͬ����Ԫ���������ҽ��������������Խ����Խ�ǿ����Mg���ȽϽ�����ǿ����������������������ˮ����ļ���ǿ����Ҳ�������ݺ�ˮ���ᷴӦ�������������׳̶ȣ���������֮�����û������ȡ�����̫�ã������������Ĥ������a���������ȽϽ�����ǿ��������û���⻯����Դ���bc
��1���ԣ�2��̼�������������CO2�����ڹ��ۻ��������ʽΪ
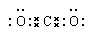 ��
����3��ͬ����Ԫ����������ԭ�Ӱ뾶��С��ͬ����Ԫ�����϶���ԭ�Ӱ뾶���������Ԣݡ��ޡ����ԭ�Ӱ뾶��С˳��ΪNa ��Al �� F��
��4���ǽ�����Խǿ����Ӧ����������ˮ��������Ծ�Խǿ���ǽ�������N��C��Si������������HNO3��H2CO3 ��H2SiO3��
��5��������Ԫ��������õĽ������ƣ��ǽ������ȣ����Է���ʽΪNaOH + HClO4 ="=" NaClO4 + H2O��
��6��ͬ����Ԫ���������ҽ��������������Խ����Խ�ǿ����Mg���ȽϽ�����ǿ����������������������ˮ����ļ���ǿ����Ҳ�������ݺ�ˮ���ᷴӦ�������������׳̶ȣ���������֮�����û������ȡ�����̫�ã������������Ĥ������a���������ȽϽ�����ǿ��������û���⻯����Դ���bc

��ϰ��ϵ�д�
�����Ŀ

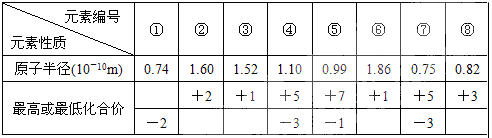
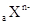 ��
�� �������ӣ�����Ӳ�ṹ��ͬ�����й�ϵʽ��ѧʽ��ȷ����
�������ӣ�����Ӳ�ṹ��ͬ�����й�ϵʽ��ѧʽ��ȷ����