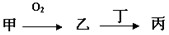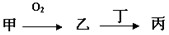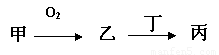��Ŀ����
��12�֣��ס��ҡ����������ʶ�����AԪ�أ�ֻ�м��ǵ��ʣ���ת����ϵ���£�
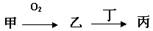
��1��������Ŀǰ���糫��������ŷŵ��������壬��Ҳ����AԪ�ء�����7�����ӵĺ���A���÷��ű�ʾΪ ������Һ�ʼ��Ե�ԭ�������ӷ���ʽ��ʾΪ ����֪���ڳ��³�ѹ�£�1mol��һ����̼�ֱ�������������Ӧ�ų��������ֱ���393.5kJ��283.0kJ����д�����������Ӧ���Ȼ�ѧ����ʽ______________ ��
��2����A�Ƿǽ���Ԫ�أ������������������������2�����������ִ�ͨѶ�п��������� ��д��һ���б��μӵġ���֤��̼�ķǽ�����ǿ��A�Ļ�ѧ����ʽ ��
��3����������������õĹ��ɽ������ʣ����ڵ�ȼ������������Ӧ�����ҡ�������ϡ���ᣬ���ҡ����Ļ�ѧ����ʽΪ ��
��12 �֣�
��1��136C��2 �֣� CO32��+
H2O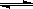 HCO3− +
OH����2 �֣�
HCO3− +
OH����2 �֣�
C(s)+CO2(g)=2CO(g) ��H= +172.5kJ/mol (2��)
��2�����ά��2 �֣��� Na2SiO3 + H2O + CO2 == H2SiO3��+ Na2CO3��2 �֣�
��3�� Fe3O4 + 8 HCl == 2FeCl 3 + FeCl 2+ 4 H2O��2 �֣�
��������

 ����Ӣ��ϵ�д�
����Ӣ��ϵ�д�
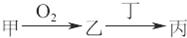 �ס��ҡ����������ʶ�����AԪ�أ�ֻ�м��ǵ��ʣ���ת����ϵ��ͼ��ʾ��
�ס��ҡ����������ʶ�����AԪ�أ�ֻ�м��ǵ��ʣ���ת����ϵ��ͼ��ʾ��