��Ŀ����
��2010?������ģ�⣩��̼������һ���ж���;��������ϵƯ������ѧʽ�ɱ�ʾΪaNa2CO3?bH2O2���ֽ�һ�������Ĺ�̼���Ʒ�ĩ����ˮ���ϡ��Һ���������м�������MnO2��ĩ����ַ�Ӧ������672mL���壨��״��������Ӧ��Ļ���ᆳ���ˡ�ϴ�Ӻ���Һ��ϴ��Һ��ϲ���ˮ���100mL��ҺA�������������Ϊ10mL��Ũ��Ϊcmol/L��ϡ�����зֱ���ε�����ҺA���ߵα�����ַ�Ӧ���ʵ���������±���ʾ��
��1������ϡ��������ʵ���Ũ�ȣ�
��2��ͨ������ȷ����̼���ƵĻ�ѧʽ��
��3����ҵ�ϳ�����������������������[w����������=
��100%]��������̼���Ʋ�Ʒ�����ӣ�13%������Ϊ�ŵ�Ʒ���ֽ�0.2gij���������Ĺ�̼������Ʒ���������ʲ���������������ԭ��Ӧ������ˮ�����Һ������15.0mL 1mol/L���ᣬ�ټ�������KI��ҡ�Ⱥ����ڰ�������ַ�Ӧ��������������Һ����0.1mol/L Na2S2O3��Һ�ζ�����ɫǡ����ʧʱ��������30.00mL���Լ����жϸ���Ʒ�Ƿ�Ϊ�ŵ�Ʒ������֪��2Na2S2O3+I2��Na2S4O6+2NaI��
| ʵ����� | �� | �� | �� |
| �μ���ҺA�����/mL | 10.0 | 20.0 | 30.0 |
| ������������/mL����״���� | 89.6 | 179.2 | 224 |
��2��ͨ������ȷ����̼���ƵĻ�ѧʽ��
��3����ҵ�ϳ�����������������������[w����������=
| 16n(H2O2) |
| m(��Ʒ) |
��������1����ͼ����֪��������ϡ������ȫ��Ӧ������̼���ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽ�Լ����ɵ������������ɼ����ϡ��������ʵ�����Ȼ�����ϡ��������ʵ���Ũ�ȣ�
��2���ֱ����̼���ƺ�˫��ˮ�����ʵ������ɣ�
��3������2I-+H2O2+2H+=I2+2H2O��2Na2S2O3+I2��Na2S4O6+2NaI��֪n��H2O2��=
��
��2���ֱ����̼���ƺ�˫��ˮ�����ʵ������ɣ�
��3������2I-+H2O2+2H+=I2+2H2O��2Na2S2O3+I2��Na2S4O6+2NaI��֪n��H2O2��=
| n(Na2S2O3) |
| 2 |
����⣺��1����ϡ��������ʵ���Ϊx��
Na2CO3+H2SO4=Na2SO4+H2O+CO2��
1 22.4L/mol
x 0.224L
=
��֮�ã�x=0.01mol��
ϡ��������ʵ���Ũ��Ϊ��C=
=
=1 mol/L��
��ϡ��������ʵ���Ũ��Ϊ1mol/L��
��2��n��O2��=
=0.03mol��n��H2O2��=0.06mol����ʵ��������ݿ�֪��10mLA��Һ�У�n��Na2CO3��=n��CO2��=0.004 mol������100mLA�к���0.04molNa2CO3��n��Na2CO3����n��H2O2��=0.04��0.06=2��3
���Ի�ѧʽΪ2Na2CO3?3H2O2����ʵ���������Ҳ�÷֣���
�𣺹�̼���ƵĻ�ѧʽΪ2Na2CO3?3H2O2��
��3������2I-+H2O2+2H+=I2+2H2O��2Na2S2O3+I2��Na2S4O6+2NaI��֪��n��H2O2��=
=
=1.5��10-3mol��
�ù�̼���ƵĻ���������������Ϊ��
=12%��13%�����Ը���Ʒ�����ŵ�Ʒ��
�𣺸���Ʒ�����ŵ�Ʒ��
Na2CO3+H2SO4=Na2SO4+H2O+CO2��
1 22.4L/mol
x 0.224L
| 1 |
| X |
| 22.4L/mol |
| 0.224L |
��֮�ã�x=0.01mol��
ϡ��������ʵ���Ũ��Ϊ��C=
| n |
| V |
| 0.01mol |
| 0.01L |
��ϡ��������ʵ���Ũ��Ϊ1mol/L��
��2��n��O2��=
| 0.672L |
| 22.4L/mol |
���Ի�ѧʽΪ2Na2CO3?3H2O2����ʵ���������Ҳ�÷֣���
�𣺹�̼���ƵĻ�ѧʽΪ2Na2CO3?3H2O2��
��3������2I-+H2O2+2H+=I2+2H2O��2Na2S2O3+I2��Na2S4O6+2NaI��֪��n��H2O2��=
| n(Na2S2O3) |
| 2 |
| 0.1mol/L��0.030L |
| 2 |
�ù�̼���ƵĻ���������������Ϊ��
| 1.5��10 -3��16 |
| 0.2 |
�𣺸���Ʒ�����ŵ�Ʒ��
���������⿼����ݻ�ѧ����ʽ�ļ��㣬�ѶȽϴ�ע��Ū�������ʽ�и����ʵ����Ĺ�ϵ�ǽ���Ĺؼ���

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
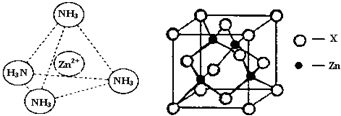
 ��R1��R2��R3����������
��R1��R2��R3����������

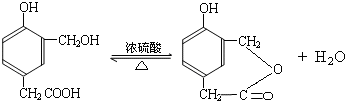
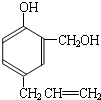
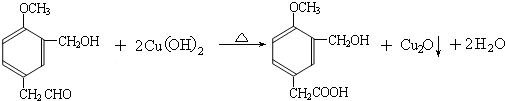
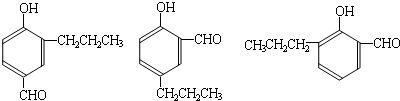
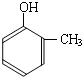 ���ĺϳ�
���ĺϳ�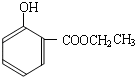 ·�ߣ��úϳ�·������ͼ��ʾ����ע����Ӧ��������
·�ߣ��úϳ�·������ͼ��ʾ����ע����Ӧ��������
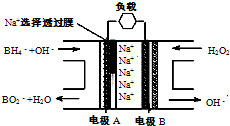 ��2010?������ģ�⣩ֱ��NaBH4/H2O2ȼ�ϵ�أ�DBFC���Ľṹ��ͼ�����������ϲ���Pt/C���������ϲ���MnO2�����йظõ�ص�˵����ȷ����
��2010?������ģ�⣩ֱ��NaBH4/H2O2ȼ�ϵ�أ�DBFC���Ľṹ��ͼ�����������ϲ���Pt/C���������ϲ���MnO2�����йظõ�ص�˵����ȷ����