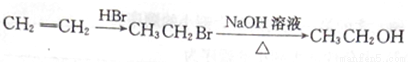��Ŀ����
OPA�������ڿ�����������е������������Ҳ���ںϳ��¿�ѪС��ۼ�ҩ���Შ�ҡ�����A�ϳ�OPA��ת����ϵ��ͼ��ʾ��
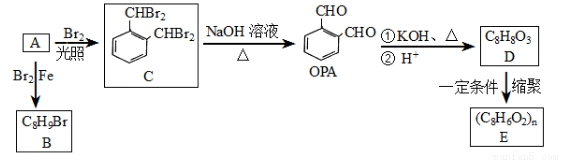
�ش��������⣺
(1)���й���A��˵����ȷ����__________(����ĸ)��
a���뱽��Ϊͬϵ�� b��������ˮ�����ӳɷ�Ӧ
c����ʹ���Ը��������Һ��ɫ d���ܷ����Ӿ۷�Ӧ���ɸ߷��ӻ�����
(2)A��B�ķ�Ӧ����Ϊ________________��
(3)OPA�Ļ�ѧ����Ϊ________________��
(4)C�����������ŵ�����Ϊ______________��
(5)E��һ�־�����߷��ӻ������D����E�Ļ�ѧ����ʽΪ_____________________��
(6)D��ͬ���칹���У����б����ұ�����ֻ������ȡ��������ˮ��������FeC13��Һ ������ɫ��Ӧ����________�֣������ܷ���������Ӧ�Һ˴Ź�������ֻ��5������ ___________(��ṹ��ʽ)��
(7)д���Լױ�Ϊԭ��(�������Լ���ѡ)�Ʊ������ᱽ�����ĺϳ�·�ߣ�__________________________________________________��
�����й�ʵ������������Ŀ�Ļ���۵�˵����ȷ����( )
ѡ�� | ���� | Ŀ�Ļ���� |
A | ��Fe(NO3)2��������ϡ����μ�KSCN��Һ���۲���Һ�Ƿ��� | ����Fe(NO3)2��Ʒ�Ƿ��ѱ����������� |
B | �������ʵ���У�Ϊ����ȡ�����ҵĽ�ȡҺ����Ҫ��к����Һ�ˮ�Ļ����1~2min | ��ҪĿ����ȥ����Һ�е������������������������� |
C | ���Թ������μ���2mLŨ���ᡢ3mL�Ҵ���2mL���ᣬ�þƾ��ƻ������� | �Ʊ��������� |
D | ��ʢ������Mg(OH)2������Թ��м�������NH4ClŨ��Һ���������ɫ�����ܽ� | NH4+��Mg(OH)2�ܽ����OH-��ϣ�����Mg(OH)2�ܽ� |
A. A B. B C. C D. D
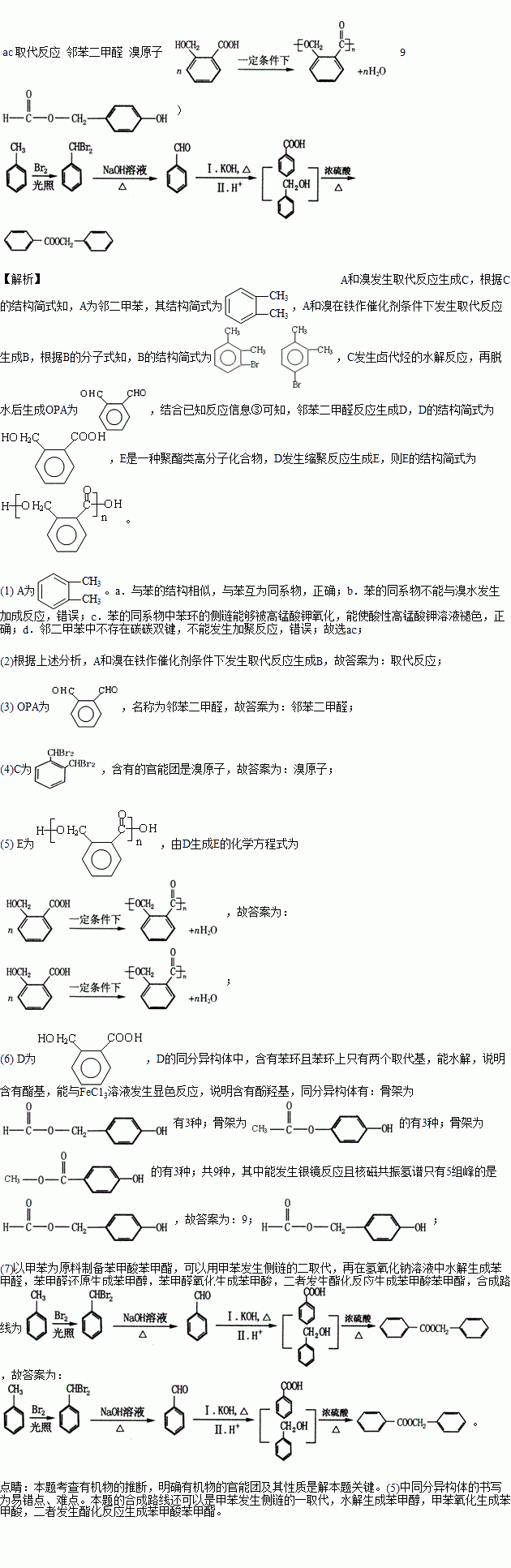

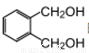 �ķ�������������ͬ���칹����_____�֡�
�ķ�������������ͬ���칹����_____�֡�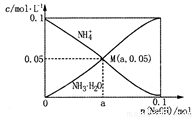
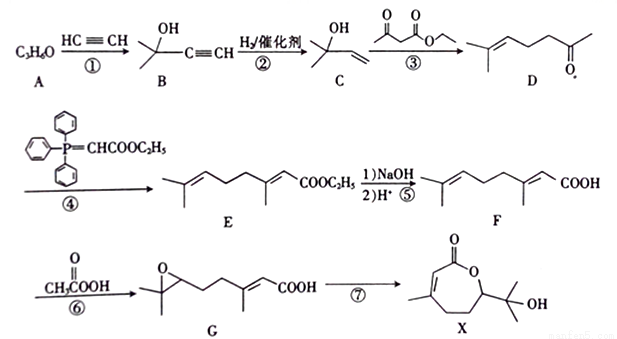
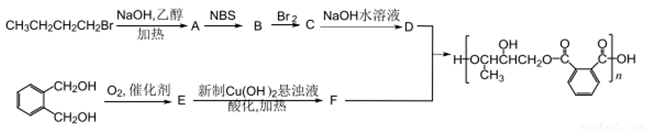
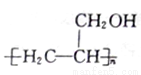 �ĺϳ�·��ͼ�����Լ����ã����ϳ�·��ʾ�����£�_______________
�ĺϳ�·��ͼ�����Լ����ã����ϳ�·��ʾ�����£�_______________