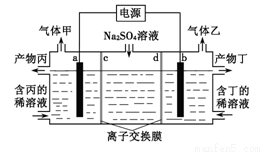
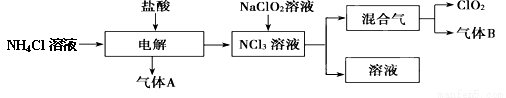
��Ŀ����
�����ƺ��⻯�ƶ������ӻ������ˮ��Ӧ�Ļ�ѧ����ʽ��δ��ƽ�����£�
Na3N+H2O��NaOH+NH3��CaH2+H2O��Ca(OH)2+H2���й����ǵ�����:
�����Ӱ뾶��Na��>N3��>H����
����ˮ��Ӧ����������ԭ��Ӧ��
����ˮ��Ӧ�����Һ���Լ��ԣ�
�������ᷴӦ��ֻ����һ���Σ�
�����ֻ������е��������Ӿ�������ͬ�ĵ��Ӳ�ṹ��
�����й�˵����ȷ���ǣ� ��
A���ڢۢܢ� B���ڢ� C���٢ۢ� D����
ú�����г����о���ͬ�¶���ƽ�ⳣ����Ͷ�ϱȼ���ֵ���� �⡣
�⡣
��֪��CO(g)��H2O(g) H2(g)��CO2(g)��ƽ�ⳣ�����¶ȵı仯���±���
H2(g)��CO2(g)��ƽ�ⳣ�����¶ȵı仯���±���
�¶�/�� | 400 | 500 | 800 |
ƽ�ⳣ��K | 9.94 | 9 | 1 |
�Իش��������⣺
��1����������Ӧ��________(����ȡ������ȡ�)��Ӧ��
��2����800 �淢��������Ӧ���Ա��е����ʵ���Ͷ����ݷ�Ӧ��������������Ӧ�����ƶ�����________(���±�����ĸ���)��
n(CO) | n(H2O) | n(H2) | n(CO2) | |
A | 1 | 5 | 2 | 3 |
B | 2 | 2 | 1 | 1 |
C | 3 | 3 | 0 | 0 |
D | 0.5 | 2 | 1 | 1 |
��3����֪��һ���¶��£�
C(s)��CO2(g) 2CO(g) ƽ�ⳣ��K1��
2CO(g) ƽ�ⳣ��K1��
C(s)��H2O(g) CO(g)��H2(g) ƽ�ⳣ��K2��
CO(g)��H2(g) ƽ�ⳣ��K2��
CO(g)��H2O(g) H2(g)��CO2(g) ƽ�ⳣ��K��
H2(g)��CO2(g) ƽ�ⳣ��K��
��K��K1��K2֮��Ĺ�ϵ��________��
��4������500 ��ʱ���У�����ʼʱCO��H2O����ʼŨ�Ⱦ�Ϊ0.020 mol��L��1���ڸ������£�COƽ��ʱ��ת����Ϊ______________��


 ���£�11.2L N2���е�������Ϊ7mol
���£�11.2L N2���е�������Ϊ7mol