��Ŀ����
8���������ȣ�ClO2��������һ�ָ�Ч�����ס���ȫ��ɱ������������������ˮ���Ʊ��������£���1��������ʳ��ˮ�Ʊ������ƣ����ڵ���ʳ��ˮ���ȳ�ȥ���е�Ca2+��Mg2+��SO42-�����ʣ��ڳ��Ӳ���ʱ��������ˮ���ȼ��������BaCl2���ѧʽ�������������ٲ������ټ��������Na2CO3��NaOH����ַ�Ӧ����һ����ȥ��
��2������������õ���ʳ��ˮ���ض������µ��õ������ƣ�NaClO3�����ٽ��������ᷴӦ����ClO2��Cl2������������ʵ�������2��1��
���� ��1���ȳ�ȥ���е�Ca2+��Mg2+��SO42-�����ʣ������Ȼ�����ȥ��������ӣ��ټ�̼���Ƴ�ȥ���������Ӽ������ӣ�NaOH��ȥþ���ӣ�
��2��NaClO3�����ᷴӦ����ClO2��Cl2��ClԪ�صĻ��ϼ���+5�۽���Ϊ+4�ۣ�ClԪ�صĻ��ϼ���-1������Ϊ0����ϵ����غ���㣮
��� �⣺��1��������ʳ��ˮ�Ʊ������ƣ����ڵ���ʳ��ˮ���ȳ�ȥ���е�Ca2+��Mg2+��SO42-�����ʣ��ڳ��Ӳ���ʱ��������ˮ���ȼ��������BaCl2����ȥ��������ӣ����������ٲ������ټ��������Na2CO3��NaOH����ַ�Ӧ����һ����ȥ���ʴ�Ϊ��BaCl2��Na2CO3��
��2��NaClO3�����ᷴӦ����ClO2��Cl2��ClԪ�صĻ��ϼ���+5�۽���Ϊ+4�ۣ�ClԪ�صĻ��ϼ���-1������Ϊ0��������ClO2��Cl2�����ʵ����ֱ�Ϊx��y��
�ɵ����غ��֪��x����5-4��=y��2��[0-��-1��]�����x��y=2��1���ʴ�Ϊ��2��1��
���� ���������ʵ��Ʊ�������������ᴿ���������з�Ӧ�ļ��㣬Ϊ��Ƶ���㣬��������֮��ķ�Ӧ��������ԭ��Ӧ�е����غ�Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
20��ijС��̽��Na2CO3��NaHCO3�����ʣ�ʵ�鲽�輰��¼���£�
�ֱ���ʢ��0.5g Na2CO3���塢0.5gNaHCO3������ձ��м���10mLˮ��20�棩�����裬�����¶� ΪT1��
���ú��º�����¶�ΪT2��
�ֱ����10mL �ܶ�ԼΪ1.1g/mL 20%�����ᣨ20�棩�����裬�����¶�T3��
�õ��±������ݣ�
�ش��������⣺
��1��NaHCO3����ˮ�Լ��ԣ���ԭ����HCO3-+H2O?H2CO3+OH-�������ӷ���ʽ��ʾ����
��2�����������ĸ����жϣ��������Na2CO3��NaHCO3�����ܷ�ȫ���ܽ��ǣ���ǡ�����
��3��������1�����ݵó���Na2CO3��������ˮ���ȣ�NaHCO3��������ˮ���ȣ�����ȡ������ȡ�����
��4����ͬѧ�����������ݵó���Na2CO3��NaHCO3�����ᷴӦ���Ƿ��ȷ�Ӧ����ͬѧ��ΪӦ������һ��ʵ�飬����������ʵ�飺�� ʢ ��10mLˮ��20�棩���ձ��м���10mL�ܶ�ԼΪ1.1g/mL20%�����ᣨ20�棩�����裬�����¶�Ϊ22.2�森
��5���������̽��������˵����ȷ����AC��
A��NaHCO3������ķ�Ӧ�����ȷ�Ӧ
B��������ϡ�������Na2CO3��NaHCO3����
C��Na2CO3��NaHCO3������ϡ���ᷴӦ�������仯�������ʵ��ܽ�������й�
��6����ͬѧΪ�ⶨһ��NaHCO3��Na2CO3��Ϲ�����NaHCO3�Ĵ��ȣ����������ʵ�鷽�������в��ܲⶨNa2CO3��NaHCO3�������Na2CO3������������C
A��ȡa�˻�����ּ��ȣ�����b��
B��ȡa�˻����������ϡ�����ַ�Ӧ�����ȡ����ɡ����գ���b�˹���
C��ȡa�˻����������ϡ�����ַ�Ӧ���ݳ������ü�ʯ�����գ�����b��
D��ȡa�˻����������Ba��OH��2��Һ��ַ�Ӧ�����ˡ�ϴ�ӡ���ɣ���b�˹��壮
����A��������ʵ�飬��ԭ�������NaHCO3����������Ϊ$\frac{84b}{31a}$��100%���ú�a��b�Ĵ���ʽ��ʾ��
�������ܽ�ȱ�
�ֱ���ʢ��0.5g Na2CO3���塢0.5gNaHCO3������ձ��м���10mLˮ��20�棩�����裬�����¶� ΪT1��
���ú��º�����¶�ΪT2��
�ֱ����10mL �ܶ�ԼΪ1.1g/mL 20%�����ᣨ20�棩�����裬�����¶�T3��
�õ��±������ݣ�
| �¶� �Լ� | T1/�� | T2/�� | T2/�� |
| Ha2CO2 | 23.3 | 20.0 | 23.7 |
| HaCO2 | 18.5 | 20.0 | 20.8 |
��1��NaHCO3����ˮ�Լ��ԣ���ԭ����HCO3-+H2O?H2CO3+OH-�������ӷ���ʽ��ʾ����
��2�����������ĸ����жϣ��������Na2CO3��NaHCO3�����ܷ�ȫ���ܽ��ǣ���ǡ�����
��3��������1�����ݵó���Na2CO3��������ˮ���ȣ�NaHCO3��������ˮ���ȣ�����ȡ������ȡ�����
��4����ͬѧ�����������ݵó���Na2CO3��NaHCO3�����ᷴӦ���Ƿ��ȷ�Ӧ����ͬѧ��ΪӦ������һ��ʵ�飬����������ʵ�飺�� ʢ ��10mLˮ��20�棩���ձ��м���10mL�ܶ�ԼΪ1.1g/mL20%�����ᣨ20�棩�����裬�����¶�Ϊ22.2�森
��5���������̽��������˵����ȷ����AC��
A��NaHCO3������ķ�Ӧ�����ȷ�Ӧ
B��������ϡ�������Na2CO3��NaHCO3����
C��Na2CO3��NaHCO3������ϡ���ᷴӦ�������仯�������ʵ��ܽ�������й�
��6����ͬѧΪ�ⶨһ��NaHCO3��Na2CO3��Ϲ�����NaHCO3�Ĵ��ȣ����������ʵ�鷽�������в��ܲⶨNa2CO3��NaHCO3�������Na2CO3������������C
A��ȡa�˻�����ּ��ȣ�����b��
B��ȡa�˻����������ϡ�����ַ�Ӧ�����ȡ����ɡ����գ���b�˹���
C��ȡa�˻����������ϡ�����ַ�Ӧ���ݳ������ü�ʯ�����գ�����b��
D��ȡa�˻����������Ba��OH��2��Һ��ַ�Ӧ�����ˡ�ϴ�ӡ���ɣ���b�˹��壮
����A��������ʵ�飬��ԭ�������NaHCO3����������Ϊ$\frac{84b}{31a}$��100%���ú�a��b�Ĵ���ʽ��ʾ��
�������ܽ�ȱ�
| �¶� �ܽ�� | 10�� | 20�� | 30�� | 40�� |
| Ha2CO2 | 12.5g | 21.5g | 39.7g | 40��g |
| HaHCO2 | 8.1g | 9.6g | 11.1g | 12.7 |
16����֪��CH4��g��+2O2��g���TCO2��g��+2H2O��l������H=-890.3kJ/mol���������������ͼ���ֱ�ȼ�պų�������֮��ԼΪ��������
| A�� | 1��3.4 | B�� | 1��1.7 | C�� | 2.6��1 | D�� | 4.6��1 |
3��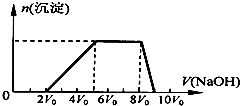 ij�����Һ�п��ܺ��е����������ʾ��
ij�����Һ�п��ܺ��е����������ʾ��
Ϊ̽����ɷ֣�����������̽��ʵ�飮
��1��̽��һ����ͬѧȡһ�����Ļ����Һ����������μ�������������Һ���������������ʵ�����n�����������������Һ�������V���Ĺ�ϵ��ͼ��ʾ��
�ٸ���Һ��һ�����е���������H+��NH4+��Al3+�����Ӧ���ʵ���Ũ��֮��Ϊ2��3��1��һ�������ڵ���������CO32-��AlO2-��
����д���������ٹ����з�����Ӧ�����ӷ���ʽAl��OH��3+OH-=AlO2$_2^-$+2H2O��
��2��̽��������ͬѧ������Һ�к��д�����Cl-��Br-��I-������1L�û����Һ��ͨ��һ������Cl2����Һ��Cl-��Br-��I-�����ʵ�����ͨ��Cl2���������״�����Ĺ�ϵ���±���ʾ��������ش��������⣺
��ԭ��Һ��Cl-��Br-��I-�����ʵ���Ũ��֮��Ϊ����������ȣ�10��15��3��
�ڵ�ͨ��Cl2������ﵽ11.2Lʱ����Һ�з������ܷ�Ӧ�����ӷ���ʽ�ɱ�ʾΪ��5Cl2+6I-+4Br-=10Cl-+3I2+2Br2��
 ij�����Һ�п��ܺ��е����������ʾ��
ij�����Һ�п��ܺ��е����������ʾ��| ���ܴ������е������� | H+��Ag+��Mg2+��Al3+��NH4+��Fe3+ |
| ���ܴ������е������� | Cl-��Br-��I-��CO32-��AlO2- |
��1��̽��һ����ͬѧȡһ�����Ļ����Һ����������μ�������������Һ���������������ʵ�����n�����������������Һ�������V���Ĺ�ϵ��ͼ��ʾ��
�ٸ���Һ��һ�����е���������H+��NH4+��Al3+�����Ӧ���ʵ���Ũ��֮��Ϊ2��3��1��һ�������ڵ���������CO32-��AlO2-��
����д���������ٹ����з�����Ӧ�����ӷ���ʽAl��OH��3+OH-=AlO2$_2^-$+2H2O��
��2��̽��������ͬѧ������Һ�к��д�����Cl-��Br-��I-������1L�û����Һ��ͨ��һ������Cl2����Һ��Cl-��Br-��I-�����ʵ�����ͨ��Cl2���������״�����Ĺ�ϵ���±���ʾ��������ش��������⣺
| Cl2���������״���� | 5.6L | 11.2L | 22.4L |
| n ��Cl-�� | 2.5mol | 3.0mol | 4.0mol |
| n ��Br-�� | 3.0mol | 2.6mol | 1.6mol |
| n ��I-�� | x mol | 0 | 0 |
�ڵ�ͨ��Cl2������ﵽ11.2Lʱ����Һ�з������ܷ�Ӧ�����ӷ���ʽ�ɱ�ʾΪ��5Cl2+6I-+4Br-=10Cl-+3I2+2Br2��
13������ʽ�뱽�����ᣨ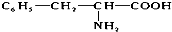 ����ͬ����ͬʱ�������������������ٱ����ϴ�������ȡ����������һ������ֱ�����ڱ����ϣ�����ϴ�������ͬ���칹����Ŀ�ǣ�������
����ͬ����ͬʱ�������������������ٱ����ϴ�������ȡ����������һ������ֱ�����ڱ����ϣ�����ϴ�������ͬ���칹����Ŀ�ǣ�������
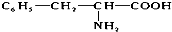 ����ͬ����ͬʱ�������������������ٱ����ϴ�������ȡ����������һ������ֱ�����ڱ����ϣ�����ϴ�������ͬ���칹����Ŀ�ǣ�������
����ͬ����ͬʱ�������������������ٱ����ϴ�������ȡ����������һ������ֱ�����ڱ����ϣ�����ϴ�������ͬ���칹����Ŀ�ǣ�������| A�� | 3 | B�� | 5 | C�� | 6 | D�� | 10 |
20��1.52gͭþ�Ͻ���ȫ�ܽ���50mL �ܶ�Ϊ1.40g/mL����������Ϊ63%��Ũ�����У��õ�NO2��N2O4�Ļ������1 120mL ����״��������Ӧ�����Һ�м���1.0mol/L NaOH��Һ������������ȫ������ʱ���õ�2.54g����������˵������ȷ���ǣ�������
| A�� | �õ�2.54 g����ʱ������NaOH��Һ�������600 mL | |
| B�� | ��Ũ������HNO3�����ʵ���Ũ����14.0 mol/L | |
| C�� | NO2��N2O4 �Ļ�������У�NO2�����������80% | |
| D�� | �úϽ���ͭ��þ�����ʵ���֮����2��1 |


 �й���ѧԺ����Ӧ�û�ѧ�о����ڼ״�ȼ�ϵ�ؼ�����������ͻ�ƣ���װ�����Ժ�����ؼ�����ʽ��أ��״�ȼ�ϵ�صĹ���ԭ����ͼ��ʾ��
�й���ѧԺ����Ӧ�û�ѧ�о����ڼ״�ȼ�ϵ�ؼ�����������ͻ�ƣ���װ�����Ժ�����ؼ�����ʽ��أ��״�ȼ�ϵ�صĹ���ԭ����ͼ��ʾ��