��Ŀ����
��������е����ⳣ�漰����ѧ֪ʶ����������������ǣ�
A�������������Ѽ�·�ˡ����������γɵ����ܽ��ж����ЧӦ
B���Ž�����¬�����Լ���Ϊ�У�����Ϊ���ɣ����������ۡ�������ָ�������ĺϽ�
C�������ݾ���ע���м�����������ʯ(KNO3)������(Na2SO4)�ķ��������Ի���֮����������������ʯҲ����������������ɫ��Ӧ
D����������������м��ء�����һ�գ���ˮ�����գ���ȡ֭�����������ص���ȡ����Ϊ��ѧ�仯
��ϰ��ϵ�д�
 ��˼ά������ҵϵ�д�
��˼ά������ҵϵ�д�
�����Ŀ


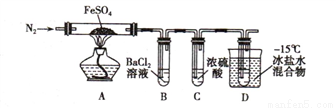
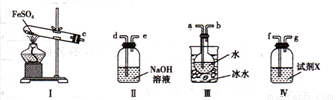
 С����̼��Ũ����Ϊ��ʼԭ�ϣ��������װ������һ��������������Ʒ�Ӧ�Ʊ��������ơ����г�װ�ú�A�м���װ�����ԣ��������Ѽ��飩
С����̼��Ũ����Ϊ��ʼԭ�ϣ��������װ������һ��������������Ʒ�Ӧ�Ʊ��������ơ����г�װ�ú�A�м���װ�����ԣ��������Ѽ��飩
 SO3
SO3 H2SO4
H2SO4 Na
Na Na2O2
Na2O2 FeSO4(aq)
FeSO4(aq)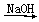 Fe(OH)2
Fe(OH)2 Fe2O
Fe2O 3
3 FeCl3(aq)
FeCl3(aq)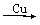 Fe
Fe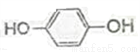 _______��
_______�� ________ (4) CH3OH________��
________ (4) CH3OH________��  ________�� (6)
________�� (6)  _________��
_________��