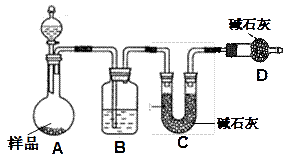
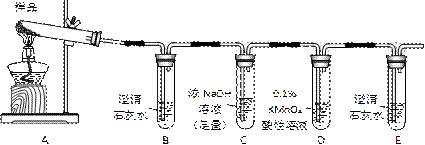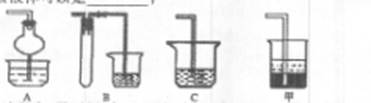��Ŀ����
�����õķ϶�ӰҺ�к���Na����[Ag(S2O3)2]3����Br�������ӡ�ij�о���ѧϰС����ͨ������ʵ�����ij���˾�ķ϶�ӰҺ����ʵ�鴦�����������е������塣
����֪��4H��+2 [Ag(S2O3)2]3�� = Ag2S��+3S��+3SO2��+SO42��+2H2O��
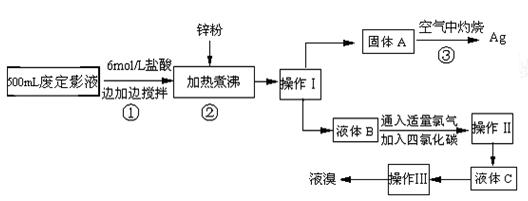
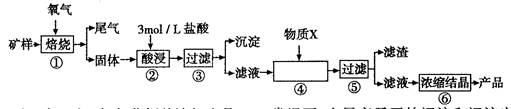
�Ų������������ ����������Ҫ����Ҫ���������� ��
�Ƽ���п�۵�Ŀ���ǽ������廯���е�����ԭ�������÷�Ӧ�����ӷ�Ӧ����ʽΪ ��
��Һ��B�г�����Br��������SO42����������Һ�д���SO42���IJ����� ��
�Ȳ����ʵ�����ʱ��Ҳ�����ü�����������ؼӸǺ������ȵķ������Ʋ������ص�Ŀ�Ŀ����� ��
��
��ͨ�������������������У�����ɫ��ѧ��Ҫ�����ڵIJ���Ϊ ��
����֪��4H��+2 [Ag(S2O3)2]3�� = Ag2S��+3S��+3SO2��+SO42��+2H2O��

�Ų������������ ����������Ҫ����Ҫ���������� ��
�Ƽ���п�۵�Ŀ���ǽ������廯���е�����ԭ�������÷�Ӧ�����ӷ�Ӧ����ʽΪ ��
��Һ��B�г�����Br��������SO42����������Һ�д���SO42���IJ����� ��
�Ȳ����ʵ�����ʱ��Ҳ�����ü�����������ؼӸǺ������ȵķ������Ʋ������ص�Ŀ�Ŀ�����
 ��
����ͨ�������������������У�����ɫ��ѧ��Ҫ�����ڵIJ���Ϊ ��
������1�֣� ��Һ©�����ձ���2�֣�
��2AgBr + Zn =" 2Ag" + Zn2+ + 2Br�� ��2�֣�
��ȡ����Һ��B��С�Թ��У��μ�BaCl2��Һ���а�ɫ����˵����SO42����2�֣�
���ṩ����ʹAg2S��ַ�Ӧ��2�֣�
���ڿ��������ղ�����SO2�ж�����Ⱦ������2�֣�
��2AgBr + Zn =" 2Ag" + Zn2+ + 2Br�� ��2�֣�
��ȡ����Һ��B��С�Թ��У��μ�BaCl2��Һ���а�ɫ����˵����SO42����2�֣�
���ṩ����ʹAg2S��ַ�Ӧ��2�֣�
���ڿ��������ղ�����SO2�ж�����Ⱦ������2�֣�
��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ