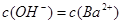��Ŀ����
A��B��C��D���ֿ������Σ�֪�������ӷֱ���Na+��Ba2+��Cu2+��Ag+ �е�ijһ�֣�
�����ӷֱ���Cl-��SO42-��CO32-��NO3- �е�ijһ�֡���������ʵ�飺
�� �������θ�ȡ�������ֱ�����ʢ��5 mL����ˮ����֧�Թ��У�ֻ��B����Һ����ɫ��
�� �ֱ���4֧�Թ��м���2 mLϡ���ᣬ����A����Һ�в�����ɫ������C����Һ���н϶����ݲ�������D����Һ����������
��1������������ʵ���ƶ��������εĻ�ѧʽ�ֱ�Ϊ��
A ��B ��C �� D ��
(2)д��ʵ�鲽������漰�������з�Ӧ�����ӷ���ʽ�� ��
�����ӷֱ���Cl-��SO42-��CO32-��NO3- �е�ijһ�֡���������ʵ�飺
�� �������θ�ȡ�������ֱ�����ʢ��5 mL����ˮ����֧�Թ��У�ֻ��B����Һ����ɫ��
�� �ֱ���4֧�Թ��м���2 mLϡ���ᣬ����A����Һ�в�����ɫ������C����Һ���н϶����ݲ�������D����Һ����������
��1������������ʵ���ƶ��������εĻ�ѧʽ�ֱ�Ϊ��
A ��B ��C �� D ��
(2)д��ʵ�鲽������漰�������з�Ӧ�����ӷ���ʽ�� ��
��1��A��AgNO3��B��CuSO4��C��Na2CO3�� D��BaCl2����1�֣�
��2��Ag++Cl-��AgCl����CO32-+2H+��CO2��+H2O����2�֣�
��2��Ag++Cl-��AgCl����CO32-+2H+��CO2��+H2O����2�֣�
�����������1��B�ε���Һ����ɫ��˵��B���к���Cu2+���ֱ���4֧�Թ��м���2mLϡ���ᣬ����A����Һ�в�����ɫ������˵��A���к���Ag+��C����Һ���н϶����ݲ�����˵��C���к���CO32-������ΪA��B��C��D�����ξ�Ϊ�������Σ���A��ΪAgNO3��C��ΪNa2CO3��B��ΪCuSO4��D��ΪBaCl2��
��2��ʵ�鲽������漰�������з�Ӧ�����ӷ���ʽ��Ag++Cl-��AgCl����CO32-+2H+��CO2��+H2O��

��ϰ��ϵ�д�
�����Ŀ
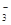 ��SO
��SO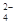 ��NH
��NH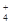 ��CO
��CO ����������Һ����ˮ�����������H+��OH������ȡ200mL����Һ����Ϊ���ȷݽ�������ʵ�飺
����������Һ����ˮ�����������H+��OH������ȡ200mL����Һ����Ϊ���ȷݽ�������ʵ�飺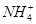 ��
��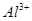 ��
�� ����
����
 ��
��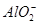 ��
��
 ��
��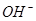 ����
����