��Ŀ����
15����A��B��C��D��E��F����Ԫ�أ�A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�ǵ縺������Ԫ�أ�C��2p�����������δ�ɶԵĵ����ӣ�Fԭ�Ӻ����������B��C���������֮�ͣ�D������Ԫ������Eͬ���ڣ�E���γɺ�ɫ����ש��ɫ����E2O�ͺ�ɫ��EO���������D��B���γ����ӻ�����侧���ṹ��ͼ��ʾ����ش��������⣮��1��A2F������Fԭ�ӵ��ӻ�������sp3��F��������FO3���ӿռ乹��Ϊƽ���������Σ�
��2����ͼ�п��Կ�����D��B�γɵ����ӻ���������ӻ����ᄃ���ܶ�Ϊag•cm-3�����������$\frac{4��78g•mo{l}^{-1}}{ag•c{m}^{-3}��{N}_{A}mo{l}^{-1}}$��д������ʽ���ɣ���
��3��E������ÿ��Eԭ����Χ���������Eԭ����ĿΪ12���ռ�������Ϊ$\frac{\sqrt{2}}{6}$�У�
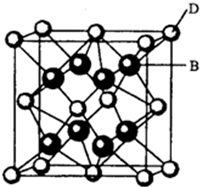
���� ��A��B��C��D��E��F����Ԫ�أ�A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ���AΪHԪ�أ�B�ǵ縺������Ԫ�أ���BΪFԪ�أ�C��2p�����������δ�ɶԵĵ����ӣ���Cԭ�Ӻ�������Ų�Ϊ1s22s22p3����CΪNԪ�أ�Fԭ�Ӻ����������B��C���������֮�ͣ���Fԭ�Ӻ��������Ϊ9+7=16����FΪSԪ�أ�E���γɺ�ɫ����ש��ɫ����E2O�ͺ�ɫ��EO�����������EΪCuԪ�أ�D��B���γ����ӻ�������ݾ����ṹ��֪��������Fԭ����ĿΪ8��Dԭ����ĿΪ8��$\frac{1}{8}$+6��$\frac{1}{2}$=4���ʻ�ѧʽΪDF2��DΪ+2�ۣ�D������Ԫ������Eͬ���ڣ����ڵ������ڣ���DΪCaԪ�أ��ݴ˽��
��� �⣺��A��B��C��D��E��F����Ԫ�أ�A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ���AΪHԪ�أ�B�ǵ縺������Ԫ�أ���BΪFԪ�أ�C��2p�����������δ�ɶԵĵ����ӣ���Cԭ�Ӻ�������Ų�Ϊ1s22s22p3����CΪNԪ�أ�Fԭ�Ӻ����������B��C���������֮�ͣ���Fԭ�Ӻ��������Ϊ9+7=16����FΪSԪ�أ�E���γɺ�ɫ����ש��ɫ����E2O�ͺ�ɫ��EO�����������EΪCuԪ�أ�D��B���γ����ӻ�������ݾ����ṹ��֪��������Fԭ����ĿΪ8��Dԭ����ĿΪ8��$\frac{1}{8}$+6��$\frac{1}{2}$=4���ʻ�ѧʽΪDF2��DΪ+2�ۣ�D������Ԫ������Eͬ���ڣ����ڵ������ڣ���DΪCaԪ�أ�
��1��H2S������Sԭ�ӵļ۲���Ӷ���=2+$\frac{6-1��2}{2}$=4��Sԭ�Ӳ�ȡsp3�ӻ���S��������SO3������Sԭ�Ӽ۲���Ӷ���=3+$\frac{6-2��3}{2}$=3��Sԭ��û�й¶Ե��ӣ���SO3Ϊƽ���������Σ�
�ʴ�Ϊ��sp3��ƽ���������Σ�
��2��D��FԪ�ؿ��γ����ӻ�������ݾ����ṹ��֪��������Fԭ����ĿΪ8��Dԭ����ĿΪ8��$\frac{1}{8}$+6��$\frac{1}{2}$=4���ʻ�ѧʽΪDF2��DΪ+2�ۣ�D������Ԫ������Eͬ���ڣ����ڵ������ڣ���DΪCaԪ�أ��ʸ����ӻ����ﻯѧʽΪCaF2���������ܶ�Ϊag•cm-3������������Ϊ$\frac{4��78g•mo{l}^{-1}}{N{\;}_{A}mo{l}^{-1}}$���ʾ��������Ϊ$\frac{\frac{4��78g•mol{\;}^{-1}}{N{\;}_{A}mol{\;}^{-1}}}{ag•cm{\;}^{-3}}$=$\frac{4��78g•mo{l}^{-1}}{ag•c{m}^{-3}��{N}_{A}mo{l}^{-1}}$��
�ʴ�Ϊ��$\frac{4��78g•mo{l}^{-1}}{ag•c{m}^{-3}��{N}_{A}mo{l}^{-1}}$��
��3��EΪCuԪ�أ�����ͭ���������������ܶѻ����ھ����Ķ�������ľ�����һ��Cuԭ�ӣ���ͭ�ľ����У�ÿ��ͭԭ����Χ���������ͭԭ�ӷֲ���ͬһ�����ĸ��ǣ���������4���������ĸ���������4�����Ӧ��4�����ܹ�12����
��ͭԭ�ӵıȽ�Ϊr�������ⳤΪ��$\frac{\sqrt{2}}{2}$��4r=2$\sqrt{2}$r�����������Ϊ����2$\sqrt{2}$r��3=16$\sqrt{2}$r3��ÿ��ͭԭ�ӵ����Ϊ��$\frac{4}{3}$��r3��ÿ�������к���4��ͭԭ�ӣ���4ͭԭ�ӵ����Ϊ��$\frac{4}{3}$��r3��4=$\frac{16}{3}$��r3������ͭ�����пռ�������=$\frac{������}{������}$��100%=$\frac{\frac{16}{3}��r{{\;}^{3}}^{\;}}{16\sqrt{2}{r}^{3}}$��100%=$\frac{\sqrt{2}}{6}$�У�
�ʴ�Ϊ��12��=$\frac{\sqrt{2}}{6}$�У�
���� ���⿼�����ӻ���������ӿռ乹�͡������ļ��㡢����ṹ�ķ�����֪ʶ����Ŀ�Ѷ��Դ��漰��֪ʶ��϶࣬��ֿ�����ѧ������ѧ֪ʶ�����������

 ������ѧ���̲���ȫ���ϵ�д�
������ѧ���̲���ȫ���ϵ�д� ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д� ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д�| A�� | m=1��n=5 | B�� | m=3��n=4 | C�� | m=5��n=1 | D�� | m=3��n=3 |
| A�� | ���ɷ��Ӿ��������һ�����й��ۼ� | |
| B�� | �ڽṹ���Ƶ�����£�ԭ�Ӿ����еĹ��ۼ�Խǿ��������۷е�Խ�� | |
| C�� | ij���Ӿ�����ۡ��е�Խ�ߣ����Ӿ����й��ۼ��ļ���Խ�� | |
| D�� | ���Ӿ�����ֻ���ڷ��Ӽ����������������κλ�ѧ�����������ۡ��е�һ��ϵ� |
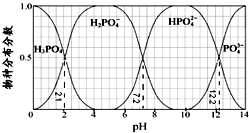
| A�� | H3PO4��Ka3=10-12.2��HPO42-��ˮ��ƽ�ⳣ��Ϊ10-6.8 | |
| B�� | Na2HPO4 ��Һ�Լ��ԣ�������Һ�м���������CaCl2 ��Һ����Һ�������� | |
| C�� | Ϊ��þ����ܴ���NaH2PO4��pHӦ������4��5.5���� | |
| D�� | pH=13ʱ����Һ�и���Ũ�ȴ�С��ϵΪ��c��Na+����c��HPO42-����c��PO43-����c��OH-����c��H+�� |
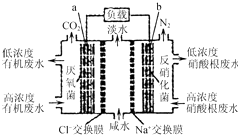 һ����������ȼ�ϵ����ˮ����ϵͳԭ����ͼ��ʾ��ͼ���л���ˮ���л������C6H10O5��ʾ�������й�˵����ȷ���ǣ�������
һ����������ȼ�ϵ����ˮ����ϵͳԭ����ͼ��ʾ��ͼ���л���ˮ���л������C6H10O5��ʾ�������й�˵����ȷ���ǣ�������| A�� | b�缫Ϊ�õ�صĸ��� | |
| B�� | b�缫������Һ��pH��С | |
| C�� | a�缫��Ӧʽ��C6H10O5-24e-+7H2O�T6CO2��+24H+ | |
| D�� | �м��ң�Na+�������ң�Cl-�������� |
| a | b | c | d | |
| A | Cu | CuSO4 | Cu��OH��2 | Cu2O |
| B | N2 | NO2 | HNO3 | NO |
| C | CH3OH | HCHO | HCOOH | HCOOCH3 |
| D | Na | Na2O2 | NaOH | NaCl |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ���ʯ | B�� | ʯī | C�� | ˮ�� | D�� | ����� |
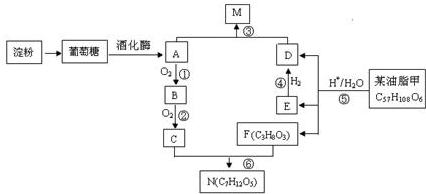
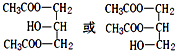 ��C��F����ͬ�ı���������������������������X��Y������Է�����X��N��Y����Y�ķ���ʽC9H14O6��
��C��F����ͬ�ı���������������������������X��Y������Է�����X��N��Y����Y�ķ���ʽC9H14O6�� C17H35COOCH2CH3+H2O��
C17H35COOCH2CH3+H2O��