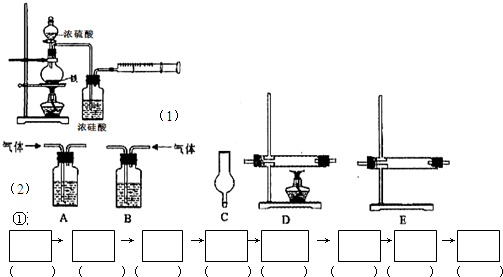
��Ŀ����
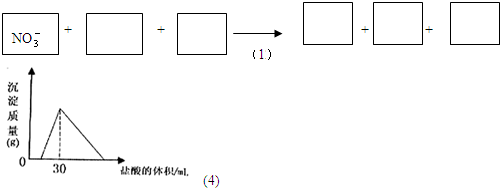
��2010?��̨��һģ����1������ר����Ϊ�����ý�������ˮ���е�NO3-ת��ΪN2���Ӷ������Ⱦ���÷�Ӧ���漰�������У�H2O��Al��OH-��Al��OH��3��NO3-��N2���뽫�����ӷֱ��������¿ո��ڣ�������ƽ�����÷�Ӧ�����У��������뱻��ԭ��Ԫ�ص����ʵ���֮��Ϊ
��2���ҹ��״��ĺ������Ժ�ˮΪ�������Һ������ܷ�ӦΪ��4Al+3O2+6H2O=4Al��OH��3����������ĵ缫��ӦʽΪ
��3����֪��4Al��s��+3O2��g��=2Al2O3��g����H=-2834.9kJ/mol
Fe2O3��s��+
C��s��=
CO2��g��+2Fe��s����H=234.1kJ/mol
C��s��+O2��g��=CO2��g����H=-393.5kJ/mol
д�������������������ȷ�Ӧ���Ȼ�ѧ����ʽ
��4����һ�������������Ͻ�����ˮ�У��Ͻ�ȫ���ܽ⣬�õ�20mol��pH=14����Һ��Ȼ��2mol/L����ζ����ɵó������������ĵ����������ϵ����ͼ����Ӧ�����в��������������Ϊ
5��3
5��3
����2���ҹ��״��ĺ������Ժ�ˮΪ�������Һ������ܷ�ӦΪ��4Al+3O2+6H2O=4Al��OH��3����������ĵ缫��ӦʽΪ
2H2O+O2+4e-=4OH-
2H2O+O2+4e-=4OH-
���������ϲ����˲��������ò���Ϊ�������ŵ����������ȶ�����������Ӧ����״�ṹ���������������ĽӴ����
�������ȶ�����������Ӧ����״�ṹ���������������ĽӴ����
����3����֪��4Al��s��+3O2��g��=2Al2O3��g����H=-2834.9kJ/mol
Fe2O3��s��+
| 3 |
| 2 |
| 3 |
| 2 |
C��s��+O2��g��=CO2��g����H=-393.5kJ/mol
д�������������������ȷ�Ӧ���Ȼ�ѧ����ʽ
2Al��s��+Fe2O3��s��=2Fe��s��=2Fe��s��+Al2O3��s����H=-593.1kJ/mol
2Al��s��+Fe2O3��s��=2Fe��s��=2Fe��s��+Al2O3��s����H=-593.1kJ/mol
����4����һ�������������Ͻ�����ˮ�У��Ͻ�ȫ���ܽ⣬�õ�20mol��pH=14����Һ��Ȼ��2mol/L����ζ����ɵó������������ĵ����������ϵ����ͼ����Ӧ�����в��������������Ϊ
2.016
2.016
L����״������
��������1�����ݽ�������ˮ���е�NO3-ת��ΪN2����֪AlԪ�صĻ��ϼ����ߣ��ڷ�Ӧ�б�������NԪ�صĻ��ϼ۽��ͣ��ڷ�Ӧ�б���ԭ�����õ����غ������㣻
��2�����ݵ���ܷ�Ӧ4Al+3O2+6H2O=4Al��OH��3��֪��Al�������������������ϵõ��ӷ�����ԭ��Ӧ�������ò������ʼ���״�ṹ�������ŵ㣻
��3��������֪�ķ�Ӧ��˹����������Ŀ�귴Ӧ�ķ�Ӧ�ȣ�����д�Ȼ�ѧ��Ӧ����ʽ��
��4������2Na+2H2O�T2NaOH+H2����2Al+2H2O+2NaOH�T2NaAlO2+3H2����NaOH+HCl�TNaCl+H2O��NaAlO2+HCl+H2O�TAl��OH��3��+NaCl�����ͼ����������������ʵ����������õ����غ����������������ʵ������������������
��2�����ݵ���ܷ�Ӧ4Al+3O2+6H2O=4Al��OH��3��֪��Al�������������������ϵõ��ӷ�����ԭ��Ӧ�������ò������ʼ���״�ṹ�������ŵ㣻
��3��������֪�ķ�Ӧ��˹����������Ŀ�귴Ӧ�ķ�Ӧ�ȣ�����д�Ȼ�ѧ��Ӧ����ʽ��
��4������2Na+2H2O�T2NaOH+H2����2Al+2H2O+2NaOH�T2NaAlO2+3H2����NaOH+HCl�TNaCl+H2O��NaAlO2+HCl+H2O�TAl��OH��3��+NaCl�����ͼ����������������ʵ����������õ����غ����������������ʵ������������������
����⣺��1���ɽ�������ˮ���е�NO3-ת��ΪN2��֪����NO3-+Al+H2O��Al��OH��3+N2+OH-�У�AlԪ�ر�������NԪ�ر���ԭ���������뱻��ԭ��Ԫ�ص����ʵ����ֱ�Ϊx��y���ɵ����غ��֪��
x����3-0��=y����5-0����
���x��y=5��3��
���������뱻��ԭ��Ԫ�ص����ʵ���֮��Ϊ5��3���ʴ�Ϊ��5��3��
��2���ɵ���ܷ�Ӧ4Al+3O2+6H2O=4Al��OH��3��֪��Al�������������������ϵõ��ӷ�����ԭ��Ӧ��
������ӦΪ2H2O+O2+4e-=4OH-��
����������ʹ�ò��������������ȶ�����������Ӧ����״�ṹ���������������ĽӴ������
�ʴ�Ϊ��2H2O+O2+4e-=4OH-���������ȶ�����������Ӧ����״�ṹ���������������ĽӴ������
��3���ɢ�4Al��s��+3O2��g��=2Al2O3��g����H=-2834.9kJ/mol��
��Fe2O3��s��+
C��s��=
CO2��g��+2Fe��s����H=234.1kJ/mol��
��C��s��+O2��g��=CO2��g����H=-393.5kJ/mol��
���ݸ�˹���ɿ�֪���١�
+��-�ۡ�
�ɵã�
2Al��s��+Fe2O3��s��=2Fe��s��=2Fe��s��+Al2O3��s����H=-593.1kJ/mol��
�ʴ�Ϊ��2Al��s��+Fe2O3��s��=2Fe��s��=2Fe��s��+Al2O3��s����H=-593.1kJ/mol��
��4����2Na+2H2O�T2NaOH+H2����2Al+2H2O+2NaOH�T2NaAlO2+3H2����NaOH+HCl�TNaCl+H2O��
NaAlO2+HCl+H2O�TAl��OH��3��+NaCl��֪��
��Ӧ��n��Na��=n��NaCl��=n��NaOH��+n��NaAlO2����
��ͼ���֪��30mL�������ɳ��������n��NaCl��=n��HCl��=0.03L��2mol/L=0.06mol��
�ɺϽ�ȫ���ܽ��õ�20mol��pH=14����Һ��n��NaOH��=0.02L��1mol/L=0.02mol��
��n��Al��=n��NaAlO2��=0.06mol-0.02mol=0.04mol��
���Ͻ���NaΪ0.06mol��AlΪ0.04mol���跴Ӧ�����в������������ʵ���Ϊn��
�ɵ����غ��֪��0.06mol��1+0.04mol��3=n��2��1��
���n=0.09mol��
�����ڱ���������Ϊ0.09mol��22.4L/mol=2.016L��
�ʴ�Ϊ��2.016��
x����3-0��=y����5-0����
���x��y=5��3��
���������뱻��ԭ��Ԫ�ص����ʵ���֮��Ϊ5��3���ʴ�Ϊ��5��3��
��2���ɵ���ܷ�Ӧ4Al+3O2+6H2O=4Al��OH��3��֪��Al�������������������ϵõ��ӷ�����ԭ��Ӧ��
������ӦΪ2H2O+O2+4e-=4OH-��
����������ʹ�ò��������������ȶ�����������Ӧ����״�ṹ���������������ĽӴ������
�ʴ�Ϊ��2H2O+O2+4e-=4OH-���������ȶ�����������Ӧ����״�ṹ���������������ĽӴ������
��3���ɢ�4Al��s��+3O2��g��=2Al2O3��g����H=-2834.9kJ/mol��
��Fe2O3��s��+
| 3 |
| 2 |
| 3 |
| 2 |
��C��s��+O2��g��=CO2��g����H=-393.5kJ/mol��
���ݸ�˹���ɿ�֪���١�
| 1 |
| 2 |
| 3 |
| 2 |
2Al��s��+Fe2O3��s��=2Fe��s��=2Fe��s��+Al2O3��s����H=-593.1kJ/mol��
�ʴ�Ϊ��2Al��s��+Fe2O3��s��=2Fe��s��=2Fe��s��+Al2O3��s����H=-593.1kJ/mol��
��4����2Na+2H2O�T2NaOH+H2����2Al+2H2O+2NaOH�T2NaAlO2+3H2����NaOH+HCl�TNaCl+H2O��
NaAlO2+HCl+H2O�TAl��OH��3��+NaCl��֪��
��Ӧ��n��Na��=n��NaCl��=n��NaOH��+n��NaAlO2����
��ͼ���֪��30mL�������ɳ��������n��NaCl��=n��HCl��=0.03L��2mol/L=0.06mol��
�ɺϽ�ȫ���ܽ��õ�20mol��pH=14����Һ��n��NaOH��=0.02L��1mol/L=0.02mol��
��n��Al��=n��NaAlO2��=0.06mol-0.02mol=0.04mol��
���Ͻ���NaΪ0.06mol��AlΪ0.04mol���跴Ӧ�����в������������ʵ���Ϊn��
�ɵ����غ��֪��0.06mol��1+0.04mol��3=n��2��1��
���n=0.09mol��
�����ڱ���������Ϊ0.09mol��22.4L/mol=2.016L��
�ʴ�Ϊ��2.016��
���������⿼��������ԭ��Ӧ��ԭ��ء���˹���ɼ��㷴Ӧ�ȡ���ѧ��Ӧ��ͼ��ļ��㣬�ѶȽϴ�֪ʶ���ۺ��Խϸߣ���4����ѧ������е��ѵ㣬ע���غ㷨�ڽ����е���Ҫ���ã�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
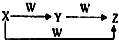 ��2010?��̨��һģ��X��Y��Z��W����ͼ��ʾ��ת����ϵ����X��Y�����Ǣ�C��CO��S��SO2��AlCl3��Al��OH��3
��2010?��̨��һģ��X��Y��Z��W����ͼ��ʾ��ת����ϵ����X��Y�����Ǣ�C��CO��S��SO2��AlCl3��Al��OH��3
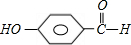 +
+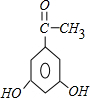
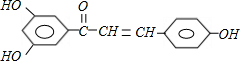 +H2O
+H2O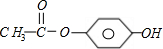
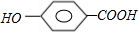 +n
+n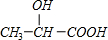
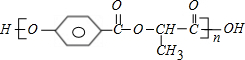 +��2n-1��H2O
+��2n-1��H2O +��2n-1��H2O
+��2n-1��H2O ��2010?��̨��һģ��[��ѧ�뼼��]��ҵ�Ͽ������÷����ᡢ̼������Ʊ�����ʯ��Na3AlF6�����乤�����£�
��2010?��̨��һģ��[��ѧ�뼼��]��ҵ�Ͽ������÷����ᡢ̼������Ʊ�����ʯ��Na3AlF6�����乤�����£�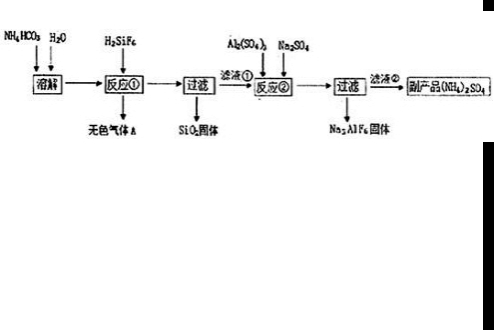
 NH3?H2O+H+��HCO3-+H2O
NH3?H2O+H+��HCO3-+H2O