��Ŀ����
���ô�������Ӧ��SO2ת��ΪSO3�ǹ�ҵ������
����Ĺؼ����裮
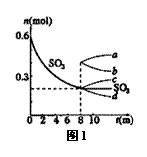 ��1��T1��ʱ����1L�ܱ������г���0.6molSO3��ͼ1��ʾSO3
��1��T1��ʱ����1L�ܱ������г���0.6molSO3��ͼ1��ʾSO3
���ʵ�����ʱ��ı仯���ߡ�
��ƽ��ʱ��SO3��ת����Ϊ (����С�����һλ) ��
T1��ʱ����Ӧ2SO2(g)+O2(g)  2SO3(g) ��ƽ�ⳣ��Ϊ �������������䣬��8minʱѹ�����������0.5L����n(SO3)�ı仯����Ϊ (����ĸ)��
2SO3(g) ��ƽ�ⳣ��Ϊ �������������䣬��8minʱѹ�����������0.5L����n(SO3)�ı仯����Ϊ (����ĸ)��
���±�Ϊ��ͬ�¶ȣ�T���£���Ӧ2SO2(g)+O2(g)  2SO3(g) ����H��0���Ļ�ѧƽ�ⳣ����K��
2SO3(g) ����H��0���Ļ�ѧƽ�ⳣ����K��
| T/�� | T2 | T3 |
| K | 20.5 | 4.68 |
�ɴ���֪���¶���͵��� ���T1������T2�� �� ��T3������
�����¶�ΪT1��ʱ�����1L���ܱ�������ͬʱ����0.2molSO2��xmolO2��0.2molSO3�������壬�ڴﵽƽ��ǰ��ҪʹSO3��Ũ�ȼ�С����x��ȡֵ��Χ�� ��
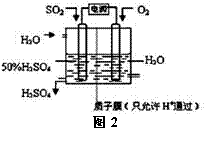 ��2����ѧ���о����õ绯ѧԭ������������¹��գ�װ����ͼ2��ʾ���ܷ�Ӧ�Ļ�ѧ����ʽΪ ���������ĵ缫��ӦʽΪ
��2����ѧ���о����õ绯ѧԭ������������¹��գ�װ����ͼ2��ʾ���ܷ�Ӧ�Ļ�ѧ����ʽΪ ���������ĵ缫��ӦʽΪ
��1���� 66.7% 1.25 c �� T2 �� 0<x<0.8
��2��2SO2+O2+2H2O=2H2SO4 SO2+2H2O -2e- = SO42-+4H+

���ñ���ʵ�����ģ������������ޣ����������Ӧʵ���һ���ǣ� ��
| ѡ�� | ʵ�����ģ�ʡ�Լг�װ�ã� | ��Ӧʵ�� |
| A | �ձ�������������ͷ�ιܡ���Һ©�� | ����ˮ�ͱ���ȥ |
| B | �ձ�������������ͷ�ιܡ���ֽ | �������ȥ���ᱵ�е�����̼�ᱵ |
| C | �ձ�������������ͷ�ιܡ�����ƿ | �ù���̼�������� |
| D | �ձ�������������ͷ�ιܡ��ƾ��� | ����ͭ��Һ��Ũ���ᾧ |
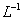 ������ζ�0.12 mol•.
������ζ�0.12 mol•.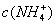 >
>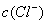 ,
,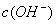 >
>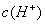 B.
B. 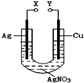
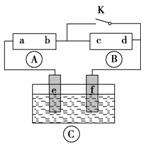
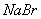 ��Һ������
��Һ������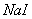
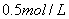 ����Һ
����Һ Ϊֱ����Դ��
Ϊֱ����Դ�� Ϊ�������Ȼ�����Һ�ͷ�̪��Һ����ֽ��
Ϊ�������Ȼ�����Һ�ͷ�̪��Һ����ֽ�� Ϊ��Ʋۡ���ͨ��·(δ�պ�K)����
Ϊ��Ʋۡ���ͨ��·(δ�պ�K)����