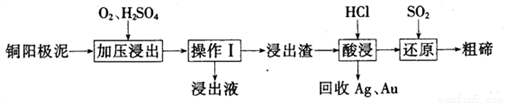
��Ŀ����
��һ������þ��ͭ��ɵĻ������뵽ϡ�����У�������ȫ�ܽ⣨���跴Ӧ�л�ԭ����ֻ��NO������Ӧ�����Һ�м���3mol/L NaOH��Һ��������ȫ��������ɳ�����������ԭ�Ͻ���������� 5.1g��������������ȷ���ǣ�������
A. ������ȫ���ܽ�ʱ���μӷ�Ӧ����������ʵ���һ����0.4mol
B. �μӷ�Ӧ�Ľ����������� 3.6g��m��9.6 g
C. �����ɵij������ﵽ���ʱ������NaOH��Һ�����V=100mL
D. ������ȫ���ܽ�ʱ�ռ���NO���������ڱ����Ϊ 2.24L
��ϰ��ϵ�д�
�����Ŀ
���ж���������ʵķ���ȫ����ȷ����
��� | ������ | ����� | ���������� | �ǵ���� |
A | Һ�� | Ư�� | Al2O3 | CO |
B | ���� | Ũ���� | Na2O2 | �Ҵ� |
C | ��ʯ�� | ���� | K2O | Cl2 |
D | ���� | ˮ���� | CaO | SO2 |
A. A B. B C. C D. D
����������͢����ȷ�����������ϵ���ǣ� ��
ѡ�� | ������ | ������ |
A | Fe3+�������� | ��KSCN��Һ���Լ���Fe3+ |
B | Ũ�����и�ʴ�� | ������Ũ�����������ʢװ |
C | Cl2�Ƿǽ������� | Cl2��ʹʪ�����ɫ������ɫ |
D | ����״̬��Al2O3���������ƶ���Al3+��O2- | �������״̬��Al2O3�Ʊ�Al |
A. A B. B C. C D. D


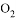 ��
�� B. �¶���ȣ�ѹǿ��ȵ�
B. �¶���ȣ�ѹǿ��ȵ�
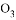 ��
�� ��
��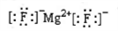
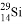
 NH4+ + 0H-
NH4+ + 0H-
 ��100%)��NaHTeO3��Һ��pH__________7(� >������<����=������
��100%)��NaHTeO3��Һ��pH__________7(� >������<����=������