��Ŀ����
ij�о���ѧϰС����ʵ���Һϳ���һ������A��
��1����������A����Է�������������100��A��C��H�����������ֱ�Ϊ��
w(C)��69.76%��w(H)��11.63%������ȫȼ�պ����ֻ��CO2��H2O��
��A��Ħ������Ϊ __________��
��2��A�ĺ˴Ź�����������ͼ��ʾ����A���Ժͽ����Ʒ�Ӧ����H2����������Cu�������±�������������ʾ���ǻ���̼̼˫�������Ľṹ���ȶ���
�����������Ϣд��A�Ľṹ��ʽ ��
��3��A��ij��ͬ���칹��B�����в���֧�����ܷ���������Ӧ��
��д��B����������Ӧ�Ļ�ѧ����ʽ
��1��86g/mol
CH3
�� CH2=CH-C-CH3
OH
��3�� CH3CH2CH2CH2CHO+ 2Ag(NH3)2OH CH3CH2CH2CH2COONH4+2Ag+3NH3+H2O
CH3CH2CH2CH2COONH4+2Ag+3NH3+H2O
���������������A����Է�������������100��A��C��H�����������ֱ�Ϊ��w(C)��69.76%��w(H)��11.63%��֪������������Ϊ1-69.76%-11.63%=18.61%������Է�������С��100�����̼�ĸ���Ϊ5������Ϊ1�����Ϊ10����A�ķ���ʽΪC5H10O�ʣ�����Ħ������Ϊ86g/mol����A���Ժͽ����Ʒ�Ӧ����H2����������Cu�������±���������֪A��Ӧ�����ǻ����������ǻ�������̼��û����ԭ�ӡ���֪A�ĽṹΪ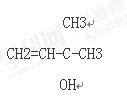 ����A��ij��ͬ���칹��B�����в���֧�����ܷ���������Ӧ��֪B�ĽṹΪCH3CH2CH2CH2CHO
����A��ij��ͬ���칹��B�����в���֧�����ܷ���������Ӧ��֪B�ĽṹΪCH3CH2CH2CH2CHO
���㣺�����л�����ӵ�ȷ���Լ��ṹ�����֪ʶ��

�ױ����ӵĶ���ȡ�����ͬ���칹�干��
| A��4�� | B��6�� | C��8�� | D��10�� |
��10�֣�������ֲ��֦Ҷ��ȡ�ľ����к������мס������ֳɷ֣�
| ����ʽ | C9H8O |
| �������� | ��ʹBr2/CCl4��ɫ |

�� ��
��1���������������ŵ�����Ϊ��������������������
��2���ɼ�ת��Ϊ���辭���й���(����ȥ������Ӧ���ز����ͬ)��

���з�Ӧ��ķ�Ӧ����Ϊ_________��Y�Ľṹ��ʽΪ_________________��
��3���������������ҷ�Ӧ��������������ѡ����ţ���
a�� ������ b�� ��ˮ c�� ̼������Һ d�� ����
��4�����ж���ͬ���칹�壬д��ͬʱ��������������ͬ���칹��ṹ��ʽ ��
a�� �����ϵ�һ�ȴ��������� b�� ����������Ӧ
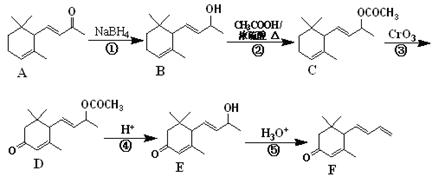
��16�֣�������E���ƽ�����ʹҩ���Ա���Ϊԭ�ϵĹ�ҵ�ϳ�·�ߣ����֣�����ͼ��
��֪�� ������д���пո�
������д���пո�
��1����Ӧ�١��ڵķ�Ӧ���� �� ��E�ķ���ʽ ��
��2��A�Ľṹ��ʽ �������й����ŵ����� ��
��3����C��Һ�м�������Ũ��ˮ��������Ӧ�Ļ�ѧ����ʽ ��
D������NaOH��Һ����ʱ��������Ӧ�Ļ�ѧ����ʽ ��
��4����ѡ�������Լ��е� �����ţ�������D��B��ˮ��Һ��
| A��FeCl3��Һ | B��FeSO4��Һ | C��NaHCO3��Һ | D��NaOH��Һ |
����һ�ַ����廯��� ������-�����ᣬ �۱�������4�ֲ�ͬ��������ԭ�ӡ�


 �������� ��
�������� �� �ɼ�дΪ
�ɼ�дΪ ������Ƭϩ�ķ��ӽṹ�ɱ�ʾΪ��
������Ƭϩ�ķ��ӽṹ�ɱ�ʾΪ��
 ����д������һ��ͬ���칹��Ľṹ��ʽ________��
����д������һ��ͬ���칹��Ľṹ��ʽ________��