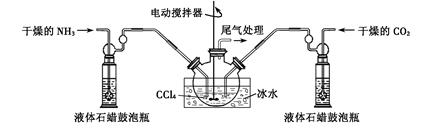
��Ŀ����
��16�֣�ͭ����ɫ�ɷ�����˾���ߵ��³������ҵ��ߵ��¡���������ҵרҵ���������ڵ������������У����ɱ���ػ����һ�����ĺ�ͭ���ϣ��磺������£���ij��ѧ��ȤС�����λͬѧ��֪��һ�����Χ�ơ��Ӻ�ͭ�����л���ͭ������˸��ԵĿ�������ͬѧ������ѧ֪ʶ�������һ���շ�����
��ͬѧ�ڲ������Ϻ��֪����ͨ����������ȵ������£�ͭ����ϡ��������Һ�з�����Ӧ������ʽΪ��2Cu+2H2SO4+O2 2CuSO4+2H2O ) ���������������һ������
2CuSO4+2H2O ) ���������������һ������
��1�������Ģ١������������У���ͭ��ͭ�Ļ������йصĻ�ѧ��Ӧ����ʽ�ֱ��ǣ�
�� ���� ��
��2���ӻ����Ƕȶ��������IJ�ͬ���ֽ��бȽϣ�����Ϊ ����ס����ҡ�)�����������������ǣ� ��
��3������Ϊ�������Ǽ����ҵķ������ڡ�����м����һ��ʱ��Ӧ�ü����Թ�������м������Ϊ����ô˵�ĵ����ǣ� ��
������������ʣ������������ʣ������ۻ���ں�ɫ��ĩ�У�����ô�����أ�
���������뷨�� ��
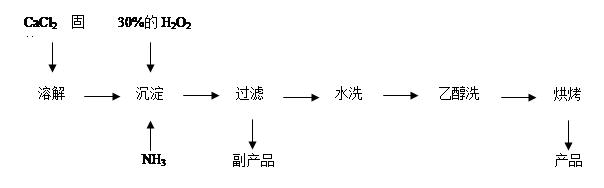
��4�������ʦ�϶���ͬѧ�ǵĻ���˼������ͬʱָ�����������һ������dz��ɫ��Һ�ᾧ��õ�һ���׳ơ��̷����Ĺ�ҵ��Ʒ�������Ӿ���Ч�档���ֱ���ŷŵ���Һ������������˷ѣ����� ��
��5���������������õ�ϡ������������Ϊ36.8%����ÿ1000mL98����Ũ���ᣨ�ܶ�Ϊ1.84g/mL�������Ƴ�����ϡ���� g����ˮ mL��ˮ���ܶ�Ϊ1.0g/mL ) ����ʵ������ϡ��Ũ����ʱ������β����ģ� ��
��1��2Cu+O2 2CuO Fe+CuSO4��FeSO4+Cu����ÿ��l�֣���2�֣�
2CuO Fe+CuSO4��FeSO4+Cu����ÿ��l�֣���2�֣�
��2���ң�l�֣�������һ�������ա��������Ⱦ���������塢�۳�����������2�֣���˼��Լ��ɣ�
��3����Cu��ȫ�û���������2�֣�������ͭ����ϴ����ϴ�ӡ������2�֣�
��4��ˮ��Ⱦ����Ⱦ����1�֣�
��5��4900��2�֣�3060��2�֣���Ũ�������ձ��ڱڻ�������ˮ�У��ӱ߽��衣��2�֣�
����������������������Ϣ���Եó�(1)�� (2)�� (3) ��(4)�Ĵ𰸡�
��1��2Cu+O2 2CuO��Fe+CuSO4��FeSO4+Cu����
2CuO��Fe+CuSO4��FeSO4+Cu����
��2���ң�������һ�������ա��������Ⱦ���������塢�۳�����������
��3����Cu��ȫ�û�������������ͭ����ϴ����ϴ�ӡ����
��4��ˮ��Ⱦ����Ⱦ��
��5�������Ƴ�����ϡ����xg����xg��36.8%=1000mL��1.84g/mL��98%��x=4900������Ũ���������Ϊ1840g���������ˮ3060g��
���㣺�����Թ�������Ϊ������������Ԫ�ؼ����������ʡ���ѧ���ӡ���ѧ�뻷��������ϵ����ѧ���㡣

 ͨ��ѧ��Ĭд����ϵ�д�
ͨ��ѧ��Ĭд����ϵ�д� ���ƽ̸�������ѡ����ĩ���100��ϵ�д�
���ƽ̸�������ѡ����ĩ���100��ϵ�д�S2Cl2�ǹ�ҵ�ϳ��õ�����ʵ�����Ʊ�S2Cl2�ķ�����2�֣�
�� CS2+3Cl2 CCl4+S2Cl2���� 2S+Cl2
CCl4+S2Cl2���� 2S+Cl2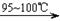 S2Cl2��
S2Cl2��
��֪S2Cl2����Ԫ����+1�ۣ�����ʽ��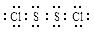 �������ȶ�����ˮ������᪻���Ӧ��һ������Ԫ�ؼ�̬���ߣ�һ���ֽ��ͣ�����Ӧ�漰�ļ������ʵ��۷е����£�
�������ȶ�����ˮ������᪻���Ӧ��һ������Ԫ�ؼ�̬���ߣ�һ���ֽ��ͣ�����Ӧ�漰�ļ������ʵ��۷е����£�
| ���� | S | CS2 | CCl4 | S2Cl2 |
| �е�/�� | 445 | 47 | 77 | 137 |
| �۵�/�� | 113 | -109 | -23 | -77 |
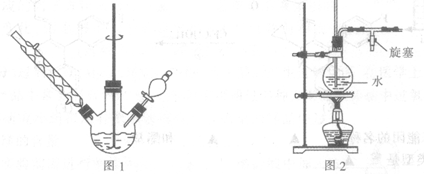
ʵ������������װ���Ʊ�S2Cl2�����ּг���������ȥ����
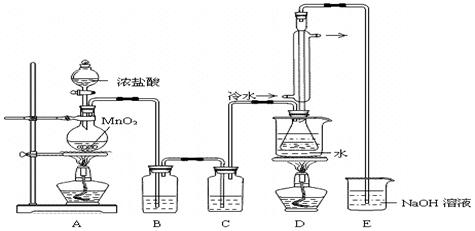
�ش��������⣺
��1��װ��B��C�в������������ƣ� ����Ӧԭ������д������ţ��� ��
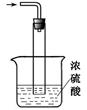
��2��ʵ���������Լ�ͨ������36.5%��Ũ��Һ������ϡ����������� ��
��3��D������������������˫�����ã�����ȴˮ��������������������������ͬ����ͼ����������ȴ��ʽ��Ӧ�������и��л�ѧ�� ʵ�顣
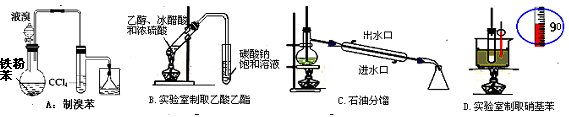
A��ʯ�ͷ��� B����ȡ�屽 C����ȡ�������� D���Ʊ���˾ƥ��
��4��Bװ����ʢ�ŵ��� ����Ӧ���������ƿ�ڻ�����з������Ʒ�ķ����� ��D�в�����ˮԡ���ȵ�ԭ���� ��
��5��A��������װ��ʱ���ź�����̨��Ӧ�ȹ̶� �����������ƣ�������װ��װ����Ϻ�Ӧ�Ƚ��� �������Լ���ʵ����ϣ�A�в��ٲ�������ʱ���ɲ��װ�á����ʱ�����ȵIJ���Ӧ���� ��
��6��ʵ������У���ȱ��Cװ�ã����ֲ�Ʒ���Dz��壬���ָ������ԭ����û�ѧ����ʽ��ʾΪ ��ʵ����ϣ�����ʣ��Ũ���ᵹ��E�ձ�����������β��������������Һ���ʱ����������������ɫ�̼�����������������������ԭ���ǣ� �������ӷ���ʽ��ʾ����
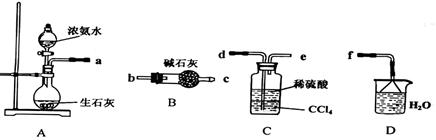

 ������ƿ�У���0.05mol/L������Һ���еζ������ı���Һ20.00mL����ش��������⣺
������ƿ�У���0.05mol/L������Һ���еζ������ı���Һ20.00mL����ش��������⣺
 NH2COONH4(s) + Q (Q > 0 )
NH2COONH4(s) + Q (Q > 0 )