��Ŀ����
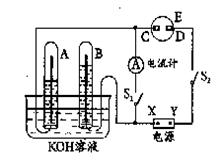
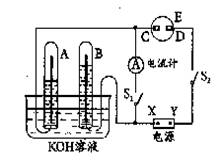
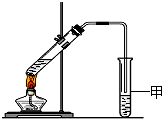
����ͼ��ʾ��ʵ��װ���У�A��ʢ��Ʒ����Һ��B��ʢ��NaOH��Һ��
 ��ش��������⣺
��ش��������⣺
(1)����A��ͨ�������Ķ����������壬A�е�������______________________��
(2)����A��ͨ���������������ټ���A��A�е���Һ______(��ܡ����ܡ�)�ָ���ԭ������ɫ��
(3)���A��ʢ���ǰ�ˮ��B��ʢ������ɫ��̪��Һ����A��B����Һ���Ϊ��ɫ�����ʱ���ְ�ˮ����______�Ժ�______�ԡ�
(4)���A��ʢ����Ũ���ᣬ����ͭ�����ȷ�Ӧ���˷�Ӧ�б���Ũ��������Ժ�______�ԡ�
(5)���A��ʢ�������ᣬ����ͭ�ۣ�Ϊ����ͭ���ܽ⣬���Լ���NaNO3����Ӧ�����ӷ���ʽΪ��3Cu��2NO��8H��===3Cu2����2NO����4H2O�����õ����ű�������ת�Ƶķ������Ŀ��
(1)��ɫ��ȥ
(2)����
(3)�ӷ��ԡ�����
(4)ǿ����
(5)3Cu��2N2e����3O��8H��===3Cu2����2NO����4H2O
����:
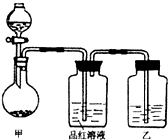
�����������Ư�����ã���ʹƷ����Һ��ɫ������Ҳ����Ư�����ã���ʹƷ����Һ��ɫ������ɫ���ټ��ȣ���Һ���ָܻ���ԭ������ɫ���õ����ű��Cu��HNO3��Ӧ�е��ӵ�ʧ�ķ������Ŀ�����£�
3Cu��2N2e����3O��8H��===3Cu2����2NO����4H2O

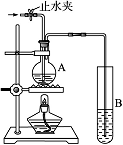
 ijͬѧ��ѧϰ�����ἰ���ε�ijЩ��������;���У���������ʵ��̽����
ijͬѧ��ѧϰ�����ἰ���ε�ijЩ��������;���У���������ʵ��̽���� CO2��+2SO2��+2H2O��
CO2��+2SO2��+2H2O��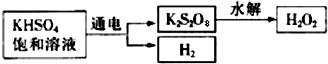
 ��1��������ֻ����̼��������Ԫ�أ��ڱ�״����Ϊ��̬���л���IJ���������������һ�����ҵ�ʯ�ͻ�����չˮƽ����ҵ�Ͽ����ñ����������������ʣ���Ӧʽ��ʾ��
��1��������ֻ����̼��������Ԫ�أ��ڱ�״����Ϊ��̬���л���IJ���������������һ�����ҵ�ʯ�ͻ�����չˮƽ����ҵ�Ͽ����ñ����������������ʣ���Ӧʽ��ʾ��