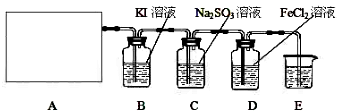
��Ŀ����
��6�֣�ȡһ֧�ྻ�Թܣ����Թ��м���0.1mol��L-1AgNO3��Һ2 mL��������������Һ
�м���������ͭ�ۣ���Ӧ�㹻��ʱ���ȡ�ϲ���Һ����һ֧�Թ��У�������Թ��еμ�
0.1 mol��L-1KI��Һ���ش��й�����ʵ����������⣺
��1�����ж���������Һ��ͭ�ķ�Ӧ������Ӧ����ȷʵ������������ ��
��2�����ж���������Һ��ͭ�ķ�Ӧ���淴ӦҲͬʱ������������ ��
��3��ʵ��������KI��Һ����KCl��Һ������ܳ���ʲô��ͬ������______
��1����Һ����ɫ �������μ�KI��Һ,��������ɫ���� (3) ������ɫ����
��������ͭ������������Cu2��������Һ������ɫ������Ӧ�ǿ���ģ�����Һ��һ������Ag����������AgI�ǻ�ɫ����������֤������KCl��Һ���������ɫ������

 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�ijУ��ѧ��ȤС��Ϊ̽������Ũ����ķ�Ӧ,�����ͼ1��ͼ2��ʾװ�ý���ʵ�顣
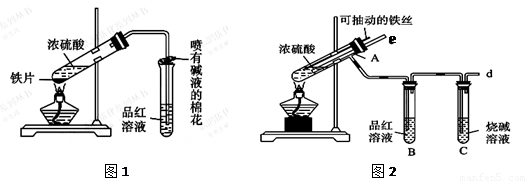
��1����˵����SO2���������ʵ�������� ��
��2��ͼ2�е�����e����Ҫ����Ϊ ��
��3������װ����ͼ2�е�NaOH��Һ������SO2β������ֹ��Ⱦ���罫�����Ϊ����KMnO4��Һ��ͬ�����ԴﵽĿ�ģ���д������KMnO4��Һ��SO2��Ӧ�Ļ�ѧ����ʽ��
��
��4���Ա�����ʵ��װ�ã����ѷ���ͼ2װ�ó����ܸ��õ������ж�����SO2��ֹ����Ⱦ�����⣬����һ���dz����Ե��ŵ㣬����Ϊ�� ��
��5����Ӧһ��ʱ���ֹͣ��Ӧ������ȴ���ý�ͷ�ι���ȡA�Թ��е���Һ���뵽����ˮ����Ϊ�����������������������ӵijɷ����������ֿ��ܣ�
��ֻ����Fe3+�� ��ֻ����Fe2+�� ����Fe3+����Fe2+��
Ϊȷ����Һ�ijɷ֣�ѡ�������Լ���
A��ϡHCl��Һ B��ϡ���� C��KSCN��Һ D������KMnO4��Һ
E��NaOH��Һ F��H2O2��Һ
�����������ص�ʵ��̽����
|
ʵ�鲽�� |
ʵ�������� |
|
1��ȡһ֧�ྻ���Թܣ��μ�1-2mL��������Һ�������Թ��еμӼ���KSCN��Һ |
��1�� ����˵��������� ��2�� ����˵����Һ�д���Fe3+������������ |
|
2��
|
�� |