��Ŀ����
������Ԫ��A��B��C��D����Ԫ�أ�ԭ���������ε�����AԪ��ԭ��2p�������2��
δ�ɶԵ��ӡ�Bԭ�ӵ�p�������3��δ�ɶԵ��ӣ�����̬�⻯����ˮ�е��ܽ����ͬ��Ԫ
�����γɵ��⻯�������C������ϼۺ���ͻ��ϼ۵Ĵ�����Ϊ4���������������
��C����������Ϊ40�����������������������������Dԭ�ӵ�һ����������3p�����
3p����ѳ�����![]()
![]()
![]()
![]()
![]()

![]()
![]()

��ش��������⣺
��1��AC2�����к���____���м�������__________���ӣ�����Է��ӡ��Ǽ��Է��ӡ���
��2�� B���⻯��Ŀռ乹��Ϊ_________��Bԭ�ӵ�ԭ�ӹ������______�ӻ������⻯����ͬ��Ԫ�����γɵ��⻯���зе���ߵ�ԭ����__________��
��3��д��A�ij�����������B���������л�Ϊ�ȵ������һ��___________
��4��CԪ�صĵ縺��_______DԪ�صĵ縺�ԣ��>������<������������һ����ѧ����ʽ��ʾ__________________________________
(1)2 �Ǽ��� (2) (3)������sp3 �����Ӽ��γ��������������ͬ������Ԫ���γɵ��⻯��е�� (4)CO2��N2O (5)< H2S +Cl2 =2HClʮS�� ![]()
![]()
![]()
![]()
![]()

![]()
![]()

����:
����AԪ��ԭ�ӵļ۵����Ų�Ϊ1s22s22p2��1s22s22p4������Ϊ̼������Bԭ�ӵļ۵����Ų�ʽΪns2np3��ΪVA��Ԫ�أ���B���⻯����ܽ�ȿ�֪BΪ��Ԫ�أ���AΪ̼��������ۺ������֮��Ϊ4����ΪVIA��Ԫ�أ�Ϊ��DԪ�ص�1�����Ӻ�3p���������˵���۵����Ų�Ϊs23p5��Ϊ��Ԫ�ء�(1)CS2�ĽṹʽΪS=C=S������2���м���ֱ���Σ��Ǽ��Է��ӡ�(2)NH3������Nԭ�ӵ�ԭ�ӹ������sp3�ӻ�������һ���ӻ����Ϊ�¶Ե���ռ�У��ռ乹��Ϊ�����Ρ�(3)̼Ԫ�صij���������CO��CO2���뵪�������ﻥΪ�ȵ��������N2O��(4)����ͬ���ڣ�ͬ����Ԫ�ص縺�ԣ���ԭ�������ĵ������������Ե縺�ԣ�C<D��

 ����������ϵ�д�
����������ϵ�д� �Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�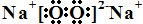
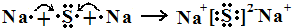
 ����������Ԫ��A��B��C��D��E��Ԫ�����ڱ��е�λ������ͼ��ʾ����ش��������⣺
����������Ԫ��A��B��C��D��E��Ԫ�����ڱ��е�λ������ͼ��ʾ����ش��������⣺