��Ŀ����
��ѧ�ҷ���ijҩ��M��������Ѫ�ܼ�������Ϊ�������������ͷų�һ�֡���ʹ���ӡ�D����������D�������ڵ�����ԭ����Ϊ�������ٻ���1998��ŵ��������ѧ��ҽѧ����������������⣺(1)��֪M����Է�������Ϊ227����C��H��O��N����Ԫ����ɣ�C��H��N��������������Ϊ15.86%��2.20%��18.50%����M�ķ���ʽ��________��D��˫ԭ�ӷ��ӣ���Է�������Ϊ30����D�ķ���ʽΪ________��
(2)��֬A������;���ɵõ�M��

ͼ8-6
ͼ�Тڵ���ʾ��
C2H5OH+HO��NO2![]() C2H5O��NO2+H2O
C2H5O��NO2+H2O
��Ӧ�ٵĻ�ѧ����ʽ��_______________________________________��
��Ӧ�ڵĻ�ѧ����ʽ��_______________________________________��
(3)C��B��������һ�������·�Ӧ���ɵĻ������Է�������Ϊ134��д��C���п��ܵĽṹ��ʽ_______________________________��
(4)����0.1 mol B�������Ľ����Ʒ�Ӧ����������________g�����ơ�
������������Ϣ�����M�ķ���ʽ����֪MΪ�������ͣ�BΪ���ͣ�AΪ֬�������������ˮ�ⷴӦ�Ļ�ѧ����ʽΪ��

C����Է�������Ϊ134������Ϊ������������ṹ��ʽ����Ϊ��
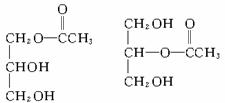
�𰸣���1��C3H5O9N NO

��4��6.9

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ




