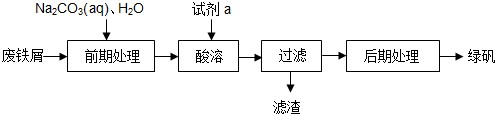
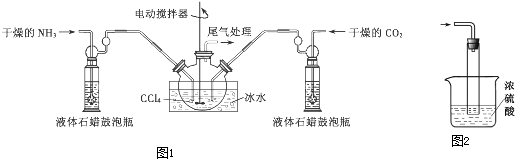
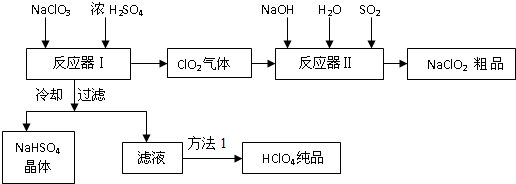
��Ŀ����
8���»�һ�л�ѧ����С����Fe��OH��3������Ʊ�ʵ���У��мס��ҡ�������ͬѧ�ֱ���������²�������ͬѧ����1mol•L-1��FeCl3��Һ�м�����NaOH��Һ��
��ͬѧ��ֱ�Ӽ��ȱ���FeCl3��Һ��
��ͬѧ����25mL��ˮ����μ���5��6��FeCl3������Һ�������������Һ�ʺ��ɫ��ֹͣ���ȣ�
�Իش��������⣺
��1�����в�����ȷ��ͬѧ�DZ�ͬѧ��
��2��֤����Fe��OH��3�������ɵ�ʵ��������ü����������Һ�����������ԵĹ�·����˵����Fe��OH��3��������д���Ƶ�Fe��OH��3����Ļ�ѧ����ʽFeCl3+3H2O����ˮ��?Fe��OH��3�����壩+3HCl��
��3���ڽ����м���������Һ������෴��ɵĽ�������ʹ������������������ͬѧ�������Ƶõ�Fe��OH��3�������ʵ�飺�ٽ���װ��U�ι��ڣ���ʯī���缫��ͨ��һ��ʱ�������������������ɫ��������Fe��OH��3�������������������������ɣ��������м��뱥��Na2SO4��Һ���������������к��ɫ�������ɣ�
���� ��1���Ʊ�������������ķ�������25ml��ˮ����μ���1��2mL FeCl3������Һ�������������Һ�ʺ��ɫ��ֹͣ���ȣ�
��2��ֻ�н�����ж����ЧӦ��������֤����IJ������Ʊ�Fe��OH��3�����ԭ�������������ˮ�⣬�Ȼ���ˮ�����������������壻
��3������ĵ�Ӿʵ��֤���˽��彺���Ǵ���ģ������м�������Ե��Ρ����ȡ������������ʹ����۳���
��� �⣺��1����ͬѧ��1mol•L-�Ȼ�����Һ�м���������NaOH��Һ���ᷢ�����ֽⷴӦ���ɺ��ɫ��������ͬѧֱ�Ӽ��ȱ���FeCl3��Һ����������ˮ�����ɺ��ɫ�������Ʊ�������������ķ�������25ml��ˮ����μ���1��2mL FeCl3������Һ�������������Һ�ʺ��ɫ��ֹͣ���ȣ�
�ʴ�Ϊ����ͬѧ��
��2��������ж����ЧӦ��������ͨ������ʱ���Ӳ���۲쵽һ�������ġ�ͨ·�����Ȼ���ˮ�����������������壬��ѧ����ʽ��FeCl3+3H2O����ˮ��?Fe��OH��3�����壩+3HCl��
�ʴ�Ϊ���ü����������Һ�����������ԵĹ�·����˵����Fe��OH��3�������ɣ�FeCl3+3H2O����ˮ��?Fe��OH��3�����壩+3HCl��
��3��������е�Ӿ���ʣ���Ӿʵ��֤���˽��彺�����㣬�������ƶ���˵��Fe��OH��3���������磬������о۳������ʣ������м�������Ե��Ρ����ȡ������������ʹ����۳���
�ʴ�Ϊ�������к��ɫ�������ɣ�
���� ���⿼��Fe��OH��3������Ʊ�֪ʶ��ע��������������ľ۳��Լ��������ܽ�ԭ���ǽ����Ĺؼ���ע�⽺���е�Ӿ���۳������ʣ���Ŀ�Ѷ��еȣ�

 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д�| A�� | ��Ũ�ȵ���Һ�е���ϡ���ᣮ�ų�����Ŀ���Na2C03��NaC03 | |
| B�� | ���ȶ���Na2C03��NaHC03 | |
| C�� | ����ʱˮ����Na2C03��NaHC03 | |
| D�� | ��Է�������Na2C03��NaHC03 |
| A�� | ���е����ӷ�Ӧ����ʽ�ǣ�Na++NH3+CO2+H2O=NaHCO3��+NH4+ | |
| B�� | ���е����ӷ�Ӧ����ʽ�ǣ�2Na++CO32-+H2O+CO2=2NaHCO3�� | |
| C�� | �����仯˵����ͬ�¶����ܽ��Na2CO3��NaHCO3��NaCl | |
| D�� | ��CaCl2��Һ�м���ŨBa��OH��2��Һ��Ҳ���������� |