��Ŀ����
ij����Һ�п��ܺ���Ag+��Fe3+��K+��Ba2+��NH4+�����ӣ���������ʵ�飺
����ô���Һ�м��������ϡ���ᣬ�а�ɫ�������ɡ�
�ڹ��ˣ�����Һ�м��������ϡ���ᣬ���а�ɫ�������ɡ�
�۹��ˣ�ȡ������Һ������2��KSCN��Һ��û�����Ե�������֡�
����ȡ����������е���Һ������NaOH��Һ����Һ�ʼ��ԣ����ȣ��ɲ���ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����塣����ʵ������ó��Ľ����У���ȷ���ǣ� ��
A. ����Һ��һ������Ba2+��Fe3+ B. ����Һ��һ������Ag+��Fe3+��NH4+
C. ����Һ��һ��������Ba2+ D. ����Һ����ȷ�������ӿ�����ɫ��Ӧ����
��ϰ��ϵ�д�
 Сѧ������ҵϵ�д�
Сѧ������ҵϵ�д� ��ʿһ��ȫͨϵ�д�
��ʿһ��ȫͨϵ�д�
�����Ŀ




 �����������ϳ�·�ߣ��Ա��ͱ���Ϊԭ�ϣ����Լ���ѡ��������Ʊ�
�����������ϳ�·�ߣ��Ա��ͱ���Ϊԭ�ϣ����Լ���ѡ��������Ʊ� �ĺϳ�·��________________��
�ĺϳ�·��________________��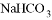 ��
��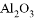 ��
�� ��
��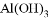 ��MgO ��Fe(OH) 3 ��CuSO4
��MgO ��Fe(OH) 3 ��CuSO4