��Ŀ����
����Ŀ���������ڹ�ҵ��Ӧ�ù㷺������������Ⱦ����ҵ��Ҫ��ԭ���ϡ�
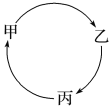
��1���Ṥҵ�̵����е�SO2��CO��������ͼ��ʾ���ۺ�ѭ�����շ��ѳ���
��֪��������ÿ���� I mol CO2��g�����ų�29��9�������������Ȼ�ѧ����ʽΪ______��
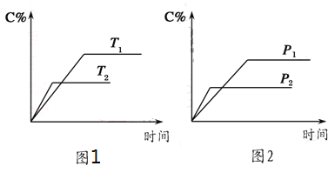
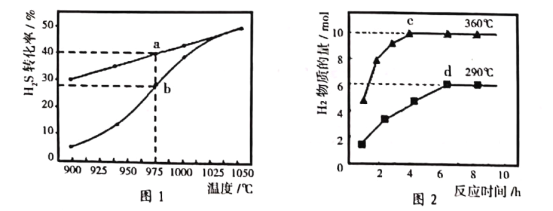
��2��H2S�ȷֽⷴӦΪ��2H2S��g��![]() 2H2��g����S2��g�����ں����ܱ������У�H2S����ʼŨ�Ⱦ�ΪcmolL��1�����Ʋ�ͬ�¶�ʹ֮�ֽ⣮��ͬʱ�����H2Sת����������ͼ1��ʾ������aΪƽ��ʱת���ʡ�bΪδƽ��ʱת���ʷֱ����¶ȵĹ�ϵ���ߡ�
2H2��g����S2��g�����ں����ܱ������У�H2S����ʼŨ�Ⱦ�ΪcmolL��1�����Ʋ�ͬ�¶�ʹ֮�ֽ⣮��ͬʱ�����H2Sת����������ͼ1��ʾ������aΪƽ��ʱת���ʡ�bΪδƽ��ʱת���ʷֱ����¶ȵĹ�ϵ���ߡ�
����975����t��ʱ��Ӧ�ﵽƽ�⣬��ƽ��ǰ��Ӧ��ƽ������v��S2����_____���ú�c��t�Ĵ���ʽ��ʾ����
����˵�����¶ȵ����ߣ�����b������a�ƽ���ԭ��______________��
��3��725��ʱ��H2S�ĵ���ƽ�ⳣ��K��1��3��10-7��Ka2��7��0��10-15����NaOH��Һ����H2S����õ�pH��10��Na2S��Һ���ڴ˹�����ˮ�ĵ���̶Ƚ�______(����������������С������������������ʱ��Һ��![]() =_____________________
=_____________________
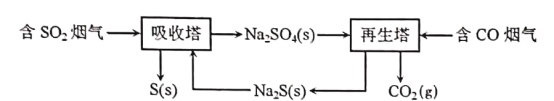
��4������Na2S��Һ��������Na2S2O3��NaOH��H2���¶�����ʱ��������Na2SO4��ͼ2�Ǻ�3 mol Na2S����Һ�ڲ�ͬ�¶��·�Ӧ������H2���ʵ����뷴Ӧʱ��Ĺ�ϵ������ͼ����ȫ��Ӧ������Na2S2O3���ʵ���������______������c������d�����㣺���������ӵ�ˮ�⣬��c����Һ��Ũ����ߵ�������Ϊ______��д��ѧʽ����S2O32�������ʵ���Ϊ_______��
���𰸡�Na2SO4(s)��4CO(g)==Na2S(s)��4CO2(g) ��H����119.6kJ��mol��1��1/4Na2SO4(s)��CO(g)==1/4Na2S(s)��CO2(g) ��H����29.9kJ��mol��1 0.2c/t mol��L��1��s��1 �¶����ߣ���Ӧ�ӿ죬�ﵽƽ������ʱ���� ���� 7.0��10��5 d SO42�� 0.5 mol
��������
��1������ͼ��ʾ���������з����ķ�Ӧ��Na2SO4��4CO=4CO2��Na2S������4molCO2ʱ�ų�������Ϊ4��29.9kJ=119.6kJ���÷�Ӧ���Ȼ�ѧ����ʽΪNa2SO4(s)��4CO(g)==Na2S(s)��4CO2(g) ��H����119.6kJ��mol��1��
��2���ٴﵽƽ��ǰ������H2S��Ũ��Ϊ0.4cmol��L��1��ͬʱ����S2Ũ��Ϊ0.4c/2mol��L��1=0.2cmol��L��1�����ݻ�ѧ��Ӧ���ʵ���ѧ����ʽ��v(S2)=0.2c/tmol/(L��s)��
�������¶����ߣ���ѧ��Ӧ�������ﵽƽ������ʱ���̣�����b������a�ƽ���
��3��NaOHΪǿ���ˮ�ĵ���������ƣ�Na2SΪǿ�������Σ�S2��ˮ��ٽ�ˮ�ĵ��룬�����NaOH��Һ����H2S��������Na2S�Ĺ����У�ˮ�ĵ���̶�����c(S2��)/c(HS��)=c(S2��)��c(H��)/[c(HS��)��c(H��)]=7.0��10��15/10��15=7.0��10��5��
��4���¶����ߣ�����Na2SO4��c���¶ȸ���d���¶ȣ�һ����S����Na2SO4���������Na2S2O3���ʵ���������d�㣻���ݷ�Ӧ����Ԫ�صĻ��ϼ����ߣ�HԪ�صĻ��ϼ۽��ͣ�������Na2S2O3�����ʵ���Ϊxmol��������Ԫ���غ㣬Na2SO4���ʵ���Ϊ(3��2x)mol�����ݵ�ʧ�����غ��غ㣬�ó�2xmol��4��(3��2x)mol��8=10mol��2�����x=0.5����Na2S2O3�����ʵ���Ϊ0.5mol��Na2SO4�����ʵ���Ϊ2mol��c����Һ��Ũ����ߵ���������SO42����S2O32�������ʵ���Ϊ0.5mol��