��Ŀ����
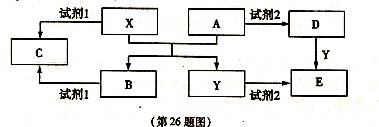
��15�֣���ͼ����ѧ��ѧ�������ʵ�ת����ϵ��ijЩ��Ӧ���������ֲ�������ȥ����A��GΪ�ճ������еij���������B��C��E��I��JΪ���壬����CΪ����ɫ���壬MΪ���ɫ���塣
��1����֪AΪ��26��Ԫ�أ���д��AԪ�������ڱ��е�λ��__________��
��2��D��G��Ӧ�Ļ�ѧ����ʽ_________________________��
��3����Ӧ�����ӷ���ʽ__________________________��
��4��F��M�IJ�������_______________��
��5���ֽ�һ�Թ�J���嵹����ˮ����һ��ʱ���ˮ������������������ʹˮ���������Թܣ�Ӧ���Թ���ͨ��һ����__________��������Ļ�ѧʽ������ʱ�Թ�����Һ��Ũ��Ϊ________mol/L�������������״�����㣩��������λ��Ч���֣���
��6����X��Y��Ϊ������Ԫ�أ�X������������C��������Ԫ������������1���Ҳ�ͬ���ڣ�Y��Xͬ���壬���й���X��Y��C��Ԫ��˵����ȷ����__________��
A��ԭ�Ӱ뾶 X��Y��C B�����Ӱ뾶 X��C��Y
C���ǽ����� X��Y��C D������������Ӧˮ�������� C��X��Y
E���⻯��е� X��Y��C
�� ��д��X���⻯����ԭ�Ӹ�����Ϊ1��1�Ļ�����ĵ���ʽ_________��
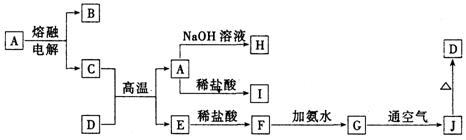
��7��D��G��Ӧ����A��ͬʱ��������һ�ֲ����ҵ������������Ҫ�ɷ�ΪAl2O3����SiO2��Fe2O3����ȡ��Ĺ����������£�
��Ϸ�ӦII���ж�������������ӣ�H+����������ǿ������˳����_________������ĸ��ţ���
A��AlO2�� B��OH�� C��SiO32��
��1���������� ��VIII�� 1��
��2��8Ai + 3Fe3O4 =����=4Al2O3 + 9Fe 2��
��3�� 3Fe + 8H+ + 2NO3��=== 3Fe2+ + 2NO��+ 4H2O 2��
��4�������ȼ������һ������أ���һ��þ������ȼþ�� 2��
��5��O2 1�� 0.045 2��
��6��B 1�� 2��
��7��BAC 2��
����:


 ��ϵ����ͼ��ʾ�����ַ�Ӧ�����������ȥ����
��ϵ����ͼ��ʾ�����ַ�Ӧ�����������ȥ����