��Ŀ����
�á�������������������⣺(1)���ԣ�H2CO3 ______ H2SiO3��H2SiO3 ______H3PO4��
(2)���ԣ�Ca(OH)
(3)��̬�⻯���ȶ��ԣ�H2O_________H2S��H2S________HCl��
(4)��ԭ�ԣ�H2O_________H2S��H2S_________HCl��
(5)���ԣ�H2SO4_________H2SO3��HClO4_________HClO��
�����ϴ��п��Թ��ɳ���
��Ԫ�صķǽ�����Խǿ��������������Ӧˮ���������Խ______________________��
��Ԫ�صĽ�����Խǿ��������������Ӧˮ����ļ���Խ______________________��
��Ԫ�ص�__________��Խǿ�����Ӧ��̬�⻯����ȶ���Խ_________________________��
�ܷǽ�����Խǿ��Ԫ�����ɵ���̬�⻯��仹ԭ��Խ______________________________��
��ͬ�ַǽ���Ԫ���γɵĺ����ᣬ�����Ԫ�ؼ�̬Խ�ߣ�������ҲԽ___________________��
����������Ԫ�������ڱ��е�λ�ã�����ͬ���ڻ�ͬ����Ԫ��ͬ�����������ʵı仯�����ƶϡ�
�𰸣�(1)�� �� (2)�� �� (3)�� �� (4)�� �� (5)�� �� ��ǿ ��ǿ �۷ǽ��� ǿ ���� ��ǿ

 ��У������Ԫͬ��ѵ��������ϵ�д�
��У������Ԫͬ��ѵ��������ϵ�д�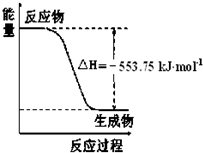 ��1����˹������Ϊ�����ܻ�ѧ������һ����ɻ��������ɣ��������̵���
��1����˹������Ϊ�����ܻ�ѧ������һ����ɻ��������ɣ��������̵���