��Ŀ����
����ʵ�������ʵ��Ŀ�IJ�һ�µ��ǣ� ��| ѡ�� | ʵ����� | ʵ��Ŀ�� |
| A | �ñ���Na2CO3��Һ�ɽ�BaSO4ת��ΪBaCO3 | ֤��Ksp�� BaCO3����Ksp�� BaSO4�� |
| B | ��PH�Ʋⶨ0.1mol/L������Һ��pH | ֤��������ˮ��Һ�в��ֵ��� |
| C | ���Թ��м�����ͬ���Ũ��Ϊ0.005mol/L�� FeCl3����Һ��0.01mol/L��KCSN��Һ���ټ��뼸�α���FeCl3��Һ | ֤������Ӧ��Ũ�ȣ�ƽ��������Ӧ�����ƶ� |
| D | ������ͨ��0.1mol/L KBr��Һ�У��ټ�������CCl4���� | ֤���ȡ���ķǽ�����ǿ�� |
A��A
B��B
C��C
D��D
���𰸡�������A�����ܵ���ʵ��ܽ�ƽ���У��ܶȻ���ĵ���������ܶȻ�С�ĵ����ת����ע��Ũ�ȵĴ�С���⣻
B�����ݴ���ĵ���̶�ȷ������ǿ����
C������Ӧ��Ũ�ȣ�������Һ��ɫ�仯�жϷ�Ӧ����
D���ǽ����ķǽ�����Խǿ���䵥�ʵ�������Խǿ�������������廯���Ƿ�Ӧȷ����������ķǽ�����ǿ����
����⣺A�������£���Na2CO3��Һ�м�����BaSO4��ĩ������BaSO4��Na2CO3��Һ�и�ŨCO32-ת��BaCO3������Ksp��BaCO3����Ksp��BaSO4������A����
B�����������ǿ����ʣ���0.1mol/L������Һ��PH=1���������1����˵�����������ᣬ�����ܸ���0.1mol/L������Һ ��pHȷ������ĵ���̶ȣ���B��ȷ��
C�������Ȼ�����Һ��Ũ�ȣ�������Һ��ɫ�仯���жϷ�Ӧ���������Һ��ɫ�����˵��ƽ��������Ӧ�����ƶ��������ܴﵽʵ��Ŀ�ģ���C��ȷ��
D���ǽ����ķǽ�����Խǿ���䵥�ʵ�������Խǿ����������������Դ����壬���������廯�ط�Ӧ�����嵥�ʣ����������Ȼ�̼��ʹ��Һ�ʳ�ɫ������������������С���壬�����ܴﵽʵ��Ŀ�ģ���D��ȷ��
��ѡA��
�������������ڳ���ʵ�������ʵ�鿼�鷶�룬ע����������ʣ������ѶȲ���ע�����֪ʶ�Ļ��ۣ�
B�����ݴ���ĵ���̶�ȷ������ǿ����
C������Ӧ��Ũ�ȣ�������Һ��ɫ�仯�жϷ�Ӧ����
D���ǽ����ķǽ�����Խǿ���䵥�ʵ�������Խǿ�������������廯���Ƿ�Ӧȷ����������ķǽ�����ǿ����
����⣺A�������£���Na2CO3��Һ�м�����BaSO4��ĩ������BaSO4��Na2CO3��Һ�и�ŨCO32-ת��BaCO3������Ksp��BaCO3����Ksp��BaSO4������A����
B�����������ǿ����ʣ���0.1mol/L������Һ��PH=1���������1����˵�����������ᣬ�����ܸ���0.1mol/L������Һ ��pHȷ������ĵ���̶ȣ���B��ȷ��
C�������Ȼ�����Һ��Ũ�ȣ�������Һ��ɫ�仯���жϷ�Ӧ���������Һ��ɫ�����˵��ƽ��������Ӧ�����ƶ��������ܴﵽʵ��Ŀ�ģ���C��ȷ��
D���ǽ����ķǽ�����Խǿ���䵥�ʵ�������Խǿ����������������Դ����壬���������廯�ط�Ӧ�����嵥�ʣ����������Ȼ�̼��ʹ��Һ�ʳ�ɫ������������������С���壬�����ܴﵽʵ��Ŀ�ģ���D��ȷ��
��ѡA��
�������������ڳ���ʵ�������ʵ�鿼�鷶�룬ע����������ʣ������ѶȲ���ע�����֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
�����Ŀ
 ijͬѧ��������װ��ʵ��ͭ��Ũ���ᡢϡ���ᷴӦ��������ͼ��ʾ��
ijͬѧ��������װ��ʵ��ͭ��Ũ���ᡢϡ���ᷴӦ��������ͼ��ʾ�� ��˵�����¾��ϴ��ر��㡱����ԭ���Ǿ��ڴ������������������ζ��������������ʵ����������Ҳ��������ͼ��ʾ��װ����ģ��ù��̣���ش��������⣺
��˵�����¾��ϴ��ر��㡱����ԭ���Ǿ��ڴ������������������ζ��������������ʵ����������Ҳ��������ͼ��ʾ��װ����ģ��ù��̣���ش��������⣺



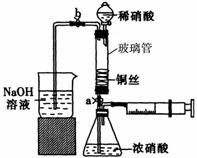 ��ϴ�Ӻ��ͭ˿���������϶������״��
��ϴ�Ӻ��ͭ˿���������϶������״��