МвДїДЪИЭ
ЎѕМвДїЎїОТ№ъїЖС§јТЧоЅьіЙ№¦єПіЙБЛКАЅзЙПКЧёцОеµЄТхАлЧУСО(N5)6(H3O)3(NH4)4ClЈЁУГ Rґъ±нЈ©ЎЈ »ШґрПВБРОКМвЈє
ЈЁ1Ј©»щМ¬µЄФЧУјЫІгµзЧУµД№мµА±нКѕКЅОЄ_____ЎЈ
ЈЁ2Ј©ВИАлЧУµД»щМ¬µзЧУЕЕІјКЅОЄ_____Ј¬УР_____ЦЦІ»Н¬ДЬј¶µДµзЧУЎЈ
ЈЁ3Ј©R ЦР HЎўNЎўO ИэЦЦФЄЛШµДµзёєРФУЙґуµЅРЎµДЛіРтКЗ_____(УГФЄЛШ·ыєЕ±нКѕ)ЎЈ
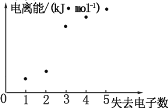
ЈЁ4Ј©ИзНј±нКѕ¶МЦЬЖЪФЄЛШ X µД»щМ¬ФЧУК§ИҐµзЧУКэУл¶ФУ¦µзАлДЬµД№ШПµЈ¬КФНЖІв X УлR ЦРµД_____(МоФЄЛШ·ыєЕ)ФЄЛШН¬ЦЬЖЪЎЈ
Ўѕґр°ёЎї![]() 1s22s22p63s23p6 5 O>N>H Cl
1s22s22p63s23p6 5 O>N>H Cl
ЎѕЅвОцЎї
(1)ёщѕЭєЛНвµзЧУЕЕІјКЅРґіцјЫІгµзЧУµД№мµА±нКѕКЅЈ»
(2)ёщѕЭВИФЧУєЛНвµзЧУЕЕІјКЅЈ¬ВИАлЧУµД»щМ¬µзЧУЕЕІјКЅЈ¬ёщѕЭµзЧУЕЕІјКЅХТіцІ»Н¬ДЬј¶µДµзЧУµДКэДїЈ»
(3)ёщѕЭ·ЗЅрКфРФµДЗїИхЈ¬ЕР¶ПіцµзёєРФµДЗїИхЈ»
(4)ёщѕЭµзАлДЬµД¶ЁТеєННјПс»ШґрЎЈ
(1)µЄКЗ7єЕФЄЛШЈ¬єЛНвµзЧУЕЕІјКЅОЄ1s22s22p3Ј¬јЫІгµзЧУµД№мµА±нКѕКЅОЄ![]() Ј»
Ј»
(2)ВИАлЧУµД»щМ¬µзЧУЕЕІјКЅОЄ1s22s22p63s23p6Ј¬УР1sЎў2sЎў2pЎў3sЎў3p5ЦЦДЬј¶І»Н¬µДµзЧУЈ»
(3)·ЗЅрКфРФOЈѕNЈѕHЈ¬ФтµзёєРФOЈѕNЈѕHЈ»
(4)¶МЦЬЖЪФЄЛШ XµДµзЧУКэі¬№э5ёцЈ¬ОЄµЪ¶ю»тµЪИэЦЬЖЪµДФЄЛШЈ¬НјПсїЙТФїґіцЈ¬К§ИҐ2ёцµзЧУєНК§ИҐ3ёцµзЧУК±µДДЬБїІоЅПґуЈ¬ЛµГчК§ИҐµДµЪИэёцµзЧУКЗОИ¶ЁЅб№№µДµзЧУЈ¬№КXµДЧоНвІгУ¦ёГУР2ёцµзЧУЈ¬XµДµзЧУКэі¬№э5ёцЈ¬XОЄГѕЈ¬УлRЦРµДClН¬Т»ЦЬЖЪЎЈ

 КАјН°ЩНЁЖЪД©ЅрѕнПµБРґр°ё
КАјН°ЩНЁЖЪД©ЅрѕнПµБРґр°ёЎѕМвДїЎїФЪ3ёціхКјОВ¶ИѕщОЄTЎжµДГЬ±ХИЭЖчЦР·ўЙъ·ґУ¦Јє2SO2(g)+O2(g)![]() 2SO3(g) (Хэ·ґУ¦·ЕИИ)ЎЈПВБРЛµ·ЁХэИ·µДКЗ( )
2SO3(g) (Хэ·ґУ¦·ЕИИ)ЎЈПВБРЛµ·ЁХэИ·µДКЗ( )
ИЭЖч±аєЕ | ИЭЖчАаРН | іхКјМе»э | ЖрКјОпЦКµДБї/mol | ЖЅєвК±SO3ОпЦКµДБї/mol | ||
SO2 | O2 | SO3 | ||||
I | єгОВєгИЭ | 1.0 L | 2 | 1 | 0 | 1.6 |
II | ѕшИИєгИЭ | 1.0 L | 2 | 1 | 0 | a |
III | єгОВєгС№ | 0.5 L | 0 | 0 | 1 | b |
A.aЈѕ1.6
B.bЈј0.8
C.ЖЅєвК±vХэ(SO2)Јєv(I)Јјv(II)
D.ИфЖрКјК±ПтИЭЖчIЦРідИл1.0 molЎЎSO2(g)Ўў0.20 mol O2(g)єН4.0ЎЎmolЎЎSO3(g)Ј¬Фт·ґУ¦Ѕ«ПтХэ·ґУ¦·ЅПтЅшРР