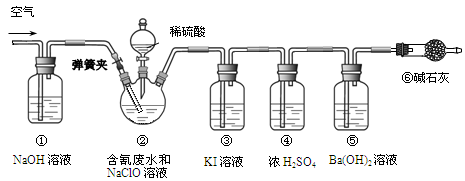
��Ŀ����
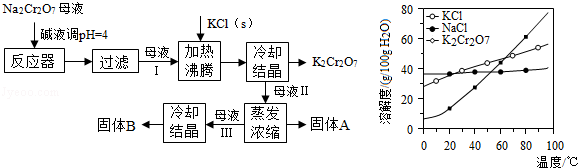
���������Ҫ�ɷֿɱ�ʾΪFeO��Cr2O3��������MgO��Al2O3��Fe2O3�����ʣ��������Ը�����Ϊԭ���Ʊ��ظ���أ�K2Cr2O7��������ͼ��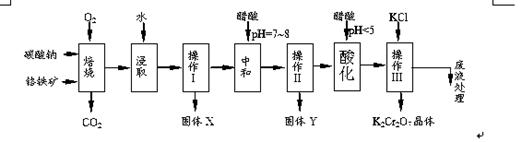
��֪����4FeO��Cr2O3+ 8Na2CO3+ 7O2 8Na2CrO4 + 2 Fe2O3 + 8CO2����
8Na2CrO4 + 2 Fe2O3 + 8CO2����
��Na2CO3 + Al2O3 2NaAlO2 + CO2������Cr2O72��+ H2O
2NaAlO2 + CO2������Cr2O72��+ H2O
 2CrO42�� + 2H+
2CrO42�� + 2H+
��������ش��������⣺
��1������X����Ҫ����_________����д��ѧʽ����Ҫ����ữ��������Һ��pH�Ƿ����4.5��Ӧ��ʹ��__________����д�������Լ����ƣ���
��2���ữ�����ô��������ҺpH<5����Ŀ����_________________________________��
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ���� �����ˡ�_______�����
��4���±���������ʵ��ܽ�����ݣ�����������Ӧ�Ļ�ѧ����ʽ�ǣ�Na2Cr2O7+2KCl ��K2Cr2O7��+2NaCl��
| ���� | �ܽ��/(g/100gˮ) | ||
| 0��C | 40��C | 80��C | |
| KCl | 28 | 40.1 | 51.3 |
| NaCl | 35.7 | 36.4 | 38 |
| K2Cr2O7 | 4.7 | 26.3 | 73 |
| Na2Cr2O7 | 163 | 215 | 376 |
�÷�Ӧ����Һ���ܷ�����������_______________��
��5������ƷY��Ҫ��������������������þ���������ܻ����P���������ʣ���ȷ����Y���������������ķ����dz�ȡn g��Ʒ���������______����д�Լ������ܽ⡢���ˡ���______����д�Լ��������ա���ȴ���������ø������m g ��������Ʒ��������������������Ϊ_________���ú�m��n�Ĵ���ʽ��ʾ����
��1��Fe2O3��MgO pH�ƻ�ȷpH��ֽ
��2��CrO42-ת��ΪCr2O72-
��3����ȴ�ᾧ��ϴ��
��4��K2Cr2O7���ܽ�ȱ�Na2Cr2O7С
��5��NaOH��Һ��ͨ�����������̼��
���������������1�����������Ҫ�ɷֿɱ�ʾΪFeO?Cr2O3��������MgO��Al2O3��Fe2O3�����ʣ�����������̼���ƣ�������ӦΪ����4FeO��Cr2O3+ 8Na2CO3+ 7O2 8Na2CrO4 + 2 Fe2O3 + 8CO2����
8Na2CrO4 + 2 Fe2O3 + 8CO2����
��Na2CO3 + Al2O3 2NaAlO2 + CO2������Cr2O72��+ H2O
2NaAlO2 + CO2������Cr2O72��+ H2O
 2CrO42�� + 2H+���������ǹ��˵õ�����XΪFe2O3��MgO��Ҫ����ữ��������Һ��pH�Ƿ����4.5����ͨpH��ֻֽ�ܲⶨ��ҺpH���������ǽ��Ʋⶨ��ȷ�ⶨ��Ҫ��pH�ƻ�ȷpH��ֽ
2CrO42�� + 2H+���������ǹ��˵õ�����XΪFe2O3��MgO��Ҫ����ữ��������Һ��pH�Ƿ����4.5����ͨpH��ֻֽ�ܲⶨ��ҺpH���������ǽ��Ʋⶨ��ȷ�ⶨ��Ҫ��pH�ƻ�ȷpH��ֽ
��2���ữ�����ô��������ҺpH��5����������ͼ�����ʵ�ת�����Ʊ�Ŀ�Ŀ�֪����Ϸ�Ӧƽ��Cr2O72-+H2O?2CrO42-+2H+�������ᣬ������Ũ������ƽ�����ƣ�������ʹCrO42-ת��ΪCr2O72-��
��3�����������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����õ����壬�ʴ�Ϊ����ȴ�ᾧ��ϴ�ӡ�
��4������ͼ�����ʵ��ܽ�ȷ����Աȣ�����������Ӧ�Ļ�ѧ����ʽ�ǣ�Na2Cr2O7+2KCl=K2Cr2O7��+2NaCl��˵��K2Cr2O7���ܽ�ȱ�Na2Cr2O7С��������������K2Cr2O7���ܽ����С�����ʴ�Ϊ��K2Cr2O7���ܽ�ȱ�Na2Cr2O7С��������������K2Cr2O7���ܽ����С����
��5������ƷY��Ҫ��������������������þ���������ܻ����P���������ʣ���ȷ����Y���������������ķ������������������������ԣ�������������Һ�ܽ������������˵õ���Һ��ͬ��ҽԺ������̼���������������������ո���õ��������������m g��������Ԫ���غ���㣬��Ʒ��������������������=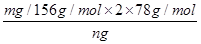 =
=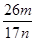 ���ʴ�Ϊ��NaOH��Һ��ͨ�����������̼��
���ʴ�Ϊ��NaOH��Һ��ͨ�����������̼��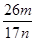 ��
��
���㣺��ѧʵ�����֪ʶ����ѧ����

����ʵ�鲻�ܴﵽĿ�ĵ���
| ѡ�� | ʵ����� | ʵ��Ŀ�� |
| A | ��5 mL 0��1 mol/L AgNO3��Һ�еμ�3��0��1 mol/L NaCl��Һ��������ɫ�������������еμ�3��0��1 mol/L��KI��Һ���л�ɫ�������� | ��֤Ksp��AgCl��> Ksp��AgI�� |
| B | ��������Ʒ��Һ�еμ�KSCN��Һ | ����FeSO4��Һ�Ƿ����� |
| C | ��������Ʒ��Һ�еμ������ữ���ٵμ�BaCl2��Һ | ����Na2SO3��Һ�Ƿ����� |
| D | ��Na2SiO3��Һ��ͨ��CO2 | ��֤�ǽ����ԣ�̼>�� |
����ʵ�鲻�ܴﵽԤ��ʵ��Ŀ�ĵ���
| ��� | ʵ������ | ʵ��Ŀ�� |
| A | ��2��5g CuSO4��5H2O�ܽ���97��5gˮ�� | ������������Ϊ1��6%��CuSO4��Һ |
| B | ��ʢ��1mL��������Һ���Թ��еμ�NaCl��Һ���������г������ɣ��������еμ�Na2S��Һ | ˵��һ�ֳ�����ת��Ϊ��һ���ܽ�ȸ�С�ij��� |
| C | �����£���pH��ֽ�ⶨŨ��Ϊ0��1mol��L��1NaClO��Һ��0��1mol��L��1CH3COONa��Һ��pH | �Ƚ�HClO��CH3COOH������ǿ�� |
| D | �����£��ֱ���2֧�Թ��м�����ͬ�������ͬŨ�ȵ�Na2S2O3��Һ���ٷֱ������ͬ�����ͬŨ�ȵ�ϡ���� | �о�Ũ�ȶԷ�Ӧ���ʵ�Ӱ�� |
(14��)
ij�о�С��Ϊ̽���������������������绯ѧ��ʴ���͵�Ӱ�����أ�����Ͼ��ȵ��������ۺ�̼��������ƿ�ײ�������ƿ��(��ͼ1)���ӽ�ͷ�ι��е��뼸�δ�����Һ��ͬʱ���������е�ѹǿ�仯��
��1�����������ʵ����Ʊ�(���в�Ҫ���ո�)��
| ��� | ʵ��Ŀ�� | ̼��/g | ����/g | ����/% |
| �� | Ϊ����ʵ�������� | 0.5 | 2.0 | 90.0 |
| �� | ����Ũ�ȵ�Ӱ�� | 0.5 | | 36.0 |
| �� | | 0.2 | 2.0 | 90.0 |
��3����С���ͼ2��0��t1ʱѹǿ����ԭ����������¼��裬������ɼ������
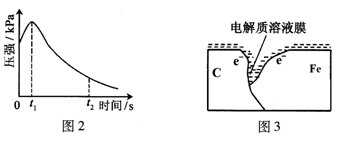
����һ���������ⸯʴ���������壻
������� ��
����
��4��Ϊ��֤����һ��ijͬѧ����˼����ռ����������Ƿ���H2�ķ��������������һ��ʵ�鷽����֤����һ��д��ʵ�鲽��ͽ��ۡ�
| ʵ�鲽��ͽ���(��Ҫ��д�����������)�� |
����������ѧ�ڷϾɵ�ػ��մ�����״�о������б�����һ��ʪ�����������������ǶԷϾɵ�ز���(����Ni(OH)2��̼�ۡ���������������)������Դ��������Ƶ�ʵ������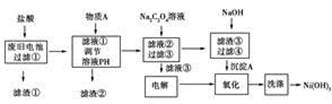
��֪����NiCl2������ˮ��Fe3����������Ni2����
����֪ʵ���¶�ʱ���ܽ�ȣ�NiC2O4��NiC2O4��H2O��NiC2O4��2H2O
�۽��������ڸ�ʵ����������ȫ������pH��
| ���� | Al3�� | Fe3�� | Ni2�� |
| pH | 5.2 | 4.1 | 9.7 |
(1)����A �������ǵ�����Һ��pH�Գ�ȥ����Fe3����Al3�����������ʺ���ΪA���ʵ���________��
A��NiO B������ C��NaOH D����ˮ
(2)�����ڵ���Ҫ�ɷ���_________________________________��
(3)д������Na2C2O4��Һ��Ӧ�Ļ�ѧ����ʽ�� _____________��
(4)д�������Һ�۵�������Ӧʽ___________________________��
��������Һ��ʱ��������������ķ�������ʪ���________��ֽ���顣
(5)�ɳ���A����Ni(OH)3�Ĺ����������������м������NaOH��Һ���г���ת��������Ӧ��ȫ����ͨ������Һ�۲��������������������д�����������̵����ӷ���ʽ��__________________��
(6)��μ���Ni(OH)3�Ƿ�ϴ�Ӹɾ���
________________________________��
�����ʵ�顱���м��ס����㡢��Լ����ɫ���ŵ㣬�۲������ĸ������ʵ�顱���г�װ��δ���������ж�����˵����ȷ���� ����������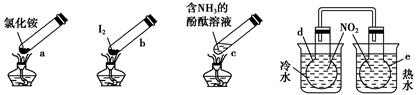
| A������ʱ��a�ϲ��ۼ��˹���NH4Cl��˵��NH4Cl�����ȶ��ԱȽϺ� |
| B������ʱ������b��I2��Ϊ��ɫ���������ϲ��־ۼ�Ϊ�Ϻ�ɫ�Ĺ��� |
| C������ʱ��c����Һ��ɫ�����ȴ���ֱ�dz |
| D��e��������ɫ��dz��d��������ɫ���� |