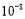��Ŀ����
����˵������ȷ����(����)
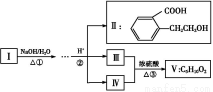
A.ʹ�ÿ�������Դ���ó��ٽ������̼����л��ܼ���ע��ԭ�ӵľ����ԡ����õ��ܺ��������ն�������ɫ��ѧ��˼��
B.����������������Ӫ������,�侧����Ҫ��������ʽ����,������Һ��pH�ɸı������ܽ��
C.ʯ���ѽ⡢ú�����������ϻ�����ˮ��þ�������绯����ά���ƻ��ȹ����ж�������ѧ�仯
D.PM2.5��ָ������ֱ��С�ڻ����2.5�Ŀ�����,Ҳ��Ϊ��ϸ��������,PM2.5�ڿ�����ֻ�γ����ܽ�
D
��������1����10-6��,1������10-9��,��1��=1 000����,����������ֱ����ΧΪ1��100����,�ʵ���2.5�Ŀ����ڿ������γɲ������ܽ�,D�����

25 ��ʱ,����ƽ�ⳣ��:
����Ļ�ѧʽ | CH3COOH | HClO | H2CO3 |
����ƽ�ⳣ��(25 ��) | 1.8��10-5 | 3.0�� | K1=4.3��10-7 K2=5.6��10-11 |
�ش���������:
(1)���ʵ���Ũ��Ϊ0.1 mol/L��������������:
a.Na2CO3,b.NaClO,c.CH3COONa,d.NaHCO3;
pH�ɴ�С��˳��������������������(����)��
(2)������0.1 mol/L��CH3COOH��Һ��ˮϡ������,���б���ʽ������һ����С������������;
A.c(H+) B.c(H+)/c(CH3COOH) C.c(H+)��c(OH-) D.c(OH-)/c(H+)
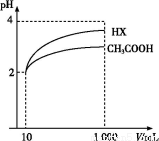
(3)���Ϊ10 mL pH=2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1 000 mL,ϡ����pH�仯��ͼ,��HX�ĵ���ƽ�ⳣ����������(������������������������С����)�����ƽ�ⳣ��;�������� ��,
ϡ�ͺ�,HX��Һ��ˮ���������c(H+)��������(������������������������С����)������Һ��ˮ���������c(H+),�������� ;
(4)25 ��ʱ,CH3COOH��CH3COONa�Ļ����Һ,����û��ҺpH=6,����Һ��c(CH3COO-)-c(Na+)=������������������������(��ȷ��ֵ)��
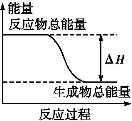
ˮú����һ�ָ�Ч����ȼ��,����Ҫ�ɷ���CO��H2,����ˮ����ͨ�����ȵ�̼�Ƶ�:C (s)+H2O(g) CO (g)+H2 (g)����H=+131 kJ��mol-1
CO (g)+H2 (g)����H=+131 kJ��mol-1
(1)T�¶���,�ĸ������о�������������Ӧ,��������̼����,�������ʵ����ʵ���Ũ�ȼ������淴Ӧ���ʹ�ϵ���±���ʾ������д������Ӧ�Ŀո�
���� ��� | c(H2O) /mol��L-1 | c(CO) /mol��L-1 | c(H2) /mol��L-1 | v����v���Ƚ� |
�� | 0.06 | 0.60 | 0.10 | v��=v�� |
�� | 0.06 | 0.50 | 0.40 | �� |
�� | 0.12 | 0.40 | 0.80 | v��<v�� |
�� | 0.12 | 0.30 | �� | v��=v�� |
������������������������,���� ��
��T�¶��¸÷�Ӧ�Ļ�ѧƽ�ⳣ��Ϊ������ ����
(2)����һ���ݻ��ɱ���ܱ����������º�ѹ��,�����м���1.0 mol̼��1.0 molˮ���� (H2O),����������Ӧ,�ﵽƽ��ʱ,�����������Ϊԭ����1.25 ����ƽ��ʱˮ������ת����Ϊ��������;��������в���a mol ̼,ˮ������ת���ʽ��������� (�� ��������������С������������)��
(3)��һ����������ˮú���ܺϳɼ״�:CO(g)+2H2(g)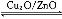 CH3OH(g)����H>0,���ϳɼ״���Ӧ��ϵ��ͨ������CO��ƽ���������� �ƶ�,��Сѹǿ��ƽ�����������ƶ�,�����¶���ƽ�������������������ƶ�(����������������������������)��
CH3OH(g)����H>0,���ϳɼ״���Ӧ��ϵ��ͨ������CO��ƽ���������� �ƶ�,��Сѹǿ��ƽ�����������ƶ�,�����¶���ƽ�������������������ƶ�(����������������������������)��