��Ŀ����
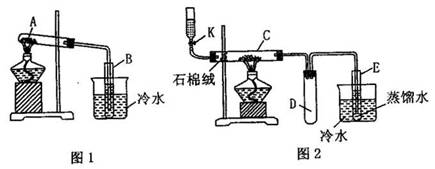
ʵ�飺�������������壨FeSO4?7H2O��������ͼ��1������ʾ�ĸ����Թ�A�����������ǿ�ȳ�ַ�Ӧ���Թ�A�еò�������ף����Թ�B�ڵõ���ɫҺ���ң�ȡ������ͼ��2���е�ʯӢ��C���в������Ӻ��������ֵ�������������K����εμ��Ҵ�ʹC����ʯ���ޣ�һ����ά״�����β��ϣ����ȶ�����ȼ�գ��������㣬�ر�K��Ȼ����ȣ����������ң����ȹ����л����ԽϿ�ؿ������ر�����K�������Ҵ���ʹ֮ͨ��ʯ��������ɹ۲쵽����ɫ�ɺ���ɫ��Ϊ��ɫ����Ӧֹͣ��ȡ�Թ�E�е�Һ��0.5mL��μ��뵽�������Ƶ�������Һ���Թ�F�У����Թܷ���ˮԡ�����ȣ����û�ۿ����Թ��ڱ��ϸ���һ������羵�Ľ�������

�ش��������⣺
��1��д����ͼ��1����ʾ�ĸ����Թ�A�з�����Ӧ�Ļ�ѧ����ʽ��
��2�����ݷ�Ӧԭ����������ͼ��1��ʵ��װ�õ���Ҫȱ���ǣ�
��3��������μ���ͼ��2����ʾװ�������ԣ�
��4���Թ�C�з�Ӧ�Ļ�ѧ����ʽΪ��
��5���Թ�D�������ǣ�
��6��д��F�Թ��з�Ӧ�Ļ�ѧ����ʽ��

�ش��������⣺
��1��д����ͼ��1����ʾ�ĸ����Թ�A�з�����Ӧ�Ļ�ѧ����ʽ��
2��FeSO4?7H2O��
Fe2O3+SO2��+SO3+14H2O
| ||
2��FeSO4?7H2O��
Fe2O3+SO2��+SO3+14H2O
��
| ||
��2�����ݷ�Ӧԭ����������ͼ��1��ʵ��װ�õ���Ҫȱ���ǣ�
SO2Ϊ������Ⱦ�ȱ��β������װ��
SO2Ϊ������Ⱦ�ȱ��β������װ��
����3��������μ���ͼ��2����ʾװ�������ԣ�
�رջ���K����E�ĵ����ܲ���ˮ�У���C�ܣ�E�������ݲ�����ֹͣ���Ⱥ�E�ܳ���һ��ˮ����˵��װ�ò�©��
�رջ���K����E�ĵ����ܲ���ˮ�У���C�ܣ�E�������ݲ�����ֹͣ���Ⱥ�E�ܳ���һ��ˮ����˵��װ�ò�©��
����4���Թ�C�з�Ӧ�Ļ�ѧ����ʽΪ��
3CH3CH2OH+Fe2O3
2Fe+3CH3CHO+3H2O
| �� |
3CH3CH2OH+Fe2O3
2Fe+3CH3CHO+3H2O
��| �� |
��5���Թ�D�������ǣ�
��ֹ����
��ֹ����
����6��д��F�Թ��з�Ӧ�Ļ�ѧ����ʽ��
CH3CHO+2Ag��NH3��2OH
CH3COONH4+2Ag��+3NH3+H2O
| �� |
CH3CHO+2Ag��NH3��2OH
CH3COONH4+2Ag��+3NH3+H2O
��| �� |
��������1���������������壨FeSO4?7H2O��������ͼ��1������ʾ�ĸ����Թ�A�����������ǿ�ȳ�ַ�Ӧ���Թ�A�еò�������ף�ͼ2װ���м��ȣ�����ɫ�ɺ���ɫ��Ϊ��ɫ���жϼ�ΪFe2O3��˵�����������������ȷֽ��Ƿ�����������ԭ��Ӧ����Ԫ�ػ��ϼ۽��ͣ���Ԫ�ػ��ϼ����ߣ����������ԭ��Ӧ�����غ�������ɲ����ж�������ϻ�ѧ����ʽ��ƽд����
��2��ͼ1װ�÷ֽ�����������˶���������Ⱦ���壬��ȱ��β������װ�ã�
��3������װ�������Լ�鷽�����лش���Ҫ������ѹǿ��ϵ�ж������Ա仯��
��4�������Ҵ��Ĵ�������Ӧԭ���ƶ�Ϊ���������Ҵ���Ӧ������ȩ��
��5��װ��D�ǰ�ȫװ�ã����������������ã�
��6��F�Թ��з�ӦΪ��ȩ��������Һ�����Ļ�ѧ����ʽ��
��2��ͼ1װ�÷ֽ�����������˶���������Ⱦ���壬��ȱ��β������װ�ã�
��3������װ�������Լ�鷽�����лش���Ҫ������ѹǿ��ϵ�ж������Ա仯��
��4�������Ҵ��Ĵ�������Ӧԭ���ƶ�Ϊ���������Ҵ���Ӧ������ȩ��
��5��װ��D�ǰ�ȫװ�ã����������������ã�
��6��F�Թ��з�ӦΪ��ȩ��������Һ�����Ļ�ѧ����ʽ��
����⣺��1���������������壨FeSO4?7H2O��������ͼ��1������ʾ�ĸ����Թ�A�����������ǿ�ȳ�ַ�Ӧ���Թ�A�еò�������ף�ͼ2װ���м��ȣ�����ɫ�ɺ���ɫ��Ϊ��ɫ���жϼ�ΪFe2O3��˵�����������������ȷֽ��Ƿ�����������ԭ��Ӧ����Ԫ�ػ��ϼ۽��ͣ���Ԫ�ػ��ϼ����ߣ����������ԭ��Ӧ�����غ�������ɲ����ж�����������ԭ���غ�͵����غ���ƽ��д���Ļ�ѧ����ʽΪ��2��FeSO4?7H2O��
Fe2O3+SO2��+SO3+14H2O��
�ʴ�Ϊ��2��FeSO4?7H2O��
Fe2O3+SO2��+SO3+14H2O��
��2�����������ֽ����ɵIJ��ﺬ��Ⱦ����������������ŷŵ������У�����װ�õ�ȱ��Ϊ��SO2Ϊ������Ⱦ�ȱ��β������װ�ã�
�ʴ�Ϊ��SO2Ϊ������Ⱦ�ȱ��β������װ�ã�
��3��װ��2����Ҫ�Ҵ���������������Ӧ������������ȩ���ռ���֤���������װ���� ��Ҫ���װ�������ԣ���Ҫ������װ���е�ѹǿ��ϵ��������鷽��Ϊ���رջ���K����E�ĵ����ܲ���ˮ�У���C�ܣ�E�������ݲ�����ֹͣ���Ⱥ�E�ܳ���һ��ˮ����˵��װ�ò�©����
�ʴ�Ϊ���رջ���K����E�ĵ����ܲ���ˮ�У���C�ܣ�E�������ݲ�����ֹͣ���Ⱥ�E�ܳ���һ��ˮ����˵��װ�ò�©����
��4���Թ�C�з�Ӧ���Ҵ��������������ķ�Ӧ����������ȩ��ˮ����Ӧ�Ļ�ѧ����ʽΪ��3CH3CH2OH+Fe2O3
2Fe+3CH3CHO+3H2O���ʴ�Ϊ��3CH3CH2OH+Fe2O3
2Fe+3CH3CHO+3H2O��
��5��װ��D�ǰ�ȫװ�ã���ֹҺ�嵹�����ʴ�Ϊ����ֹ������
��6���Թ�E�е�Һ��0.5mL��μ��뵽�������Ƶ�������Һ���Թ�F�У����Թܷ���ˮԡ�����ȣ����û�ۿ����Թ��ڱ��ϸ���һ������羵�Ľ�������֤����������ȩ����ȩ��������Һ��Ӧ����������Ӧ����Ӧ�Ļ�ѧ����ʽΪ��CH3CHO+2Ag��NH3��2OH
CH3COONH4+2Ag��+3NH3+H2O��
�ʴ�Ϊ��CH3CHO+2Ag��NH3��2OH
CH3COONH4+2Ag��+3NH3+H2O��
| ||
�ʴ�Ϊ��2��FeSO4?7H2O��
| ||
��2�����������ֽ����ɵIJ��ﺬ��Ⱦ����������������ŷŵ������У�����װ�õ�ȱ��Ϊ��SO2Ϊ������Ⱦ�ȱ��β������װ�ã�
�ʴ�Ϊ��SO2Ϊ������Ⱦ�ȱ��β������װ�ã�
��3��װ��2����Ҫ�Ҵ���������������Ӧ������������ȩ���ռ���֤���������װ���� ��Ҫ���װ�������ԣ���Ҫ������װ���е�ѹǿ��ϵ��������鷽��Ϊ���رջ���K����E�ĵ����ܲ���ˮ�У���C�ܣ�E�������ݲ�����ֹͣ���Ⱥ�E�ܳ���һ��ˮ����˵��װ�ò�©����
�ʴ�Ϊ���رջ���K����E�ĵ����ܲ���ˮ�У���C�ܣ�E�������ݲ�����ֹͣ���Ⱥ�E�ܳ���һ��ˮ����˵��װ�ò�©����
��4���Թ�C�з�Ӧ���Ҵ��������������ķ�Ӧ����������ȩ��ˮ����Ӧ�Ļ�ѧ����ʽΪ��3CH3CH2OH+Fe2O3
| �� |
| �� |
��5��װ��D�ǰ�ȫװ�ã���ֹҺ�嵹�����ʴ�Ϊ����ֹ������
��6���Թ�E�е�Һ��0.5mL��μ��뵽�������Ƶ�������Һ���Թ�F�У����Թܷ���ˮԡ�����ȣ����û�ۿ����Թ��ڱ��ϸ���һ������羵�Ľ�������֤����������ȩ����ȩ��������Һ��Ӧ����������Ӧ����Ӧ�Ļ�ѧ����ʽΪ��CH3CHO+2Ag��NH3��2OH
| �� |
�ʴ�Ϊ��CH3CHO+2Ag��NH3��2OH
| �� |
���������⿼��������ת����ϵ��Ӧ�ã��������ʵ�Ӧ�ã�ʵ��װ�÷�����ʵ�����������Ӧ�ã���Ҫ�����������ֽ������жϣ������������֤������Ĵ����ã��Ҵ��Ĵ�������Ӧ����ȩ��������Ӧ��������Ӧ�����ʵ��ۺ�Ӧ�ã���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д�
�����Ŀ