��Ŀ����
����Ŀ��һ���¶��£���2L���ܱ������м���4mol A��6mol B���������·�Ӧ��2A��g��+3B��g��![]() 4C��g��+D��g����Ӧ10min���ƽ�⣬��ʱD��Ũ��Ϊ0.5mol/L������˵����ȷ����
4C��g��+D��g����Ӧ10min���ƽ�⣬��ʱD��Ũ��Ϊ0.5mol/L������˵����ȷ����
A. ǰ10min��Ӧ��ƽ������V��C��=0.10mol/L��min
B. ��Ӧ��ƽ��ʱB��ƽ��Ũ����1.5mol/L
C. �����½���Ӧ���������Сһ�룬��D��ƽ��Ũ��С��1.0mol/L
D. 10min���������м���A�������¶Ȳ�������ƽ��ʱA��ת���ʴ���50%
���𰸡�B
��������
һ���¶��£���2L���ܱ������м���4mol A��6mol B����Ӧ10min��ﵽƽ�⣬��ʱD��Ũ����0.5molL-1����
2A��g��+3B��g��4C��g��+D��g��
��ʼŨ�ȣ�mol/L����2 3 0 0
�仯Ũ�ȣ�mol/L����1 1.5 2 0.5
ƽ��Ũ�ȣ�mol/L����1 1.5 2 0.5
A��ǰ10 min�ڷ�Ӧ��ƽ����������C��=![]() =0.2 molL-1min-1��ѡ��A����
=0.2 molL-1min-1��ѡ��A����
B�������������֪����Ӧ��ƽ��ʱc��B��=1.5 molL-1��ѡ��B��ȷ��
C�������£���ѹǿ��Ϊԭ����2�������������Сһ�룬��Ӧ��������������䣬ƽ�ⲻ�ƶ���D��Ũ��Ϊԭƽ��2������D��Ũ�ȵ���1 molL-1��ѡ��C����
D��ԭƽ����A��ת����Ϊ![]() ��100%=50%��10 min���������м���A���ٴδ�ƽ��ʱ��A��ת����С��ԭƽ�⣬���ٴδ�ƽ��ʱ��A��ת����һ��С��50%��ѡ��D����
��100%=50%��10 min���������м���A���ٴδ�ƽ��ʱ��A��ת����С��ԭƽ�⣬���ٴδ�ƽ��ʱ��A��ת����һ��С��50%��ѡ��D����
��ѡB��

����Ŀ����֪��ѧ��Ӧ����Fe��s��+CO2��g��![]() FeO��s��+CO��g������ƽ�ⳣ��ΪK1����ѧ��Ӧ����Fe��s��+H2O��g��
FeO��s��+CO��g������ƽ�ⳣ��ΪK1����ѧ��Ӧ����Fe��s��+H2O��g��![]() FeO��s��+H2��g������ƽ�ⳣ��ΪK2�����¶�973K��1173K����£�K1��K2��ֵ�ֱ����£�
FeO��s��+H2��g������ƽ�ⳣ��ΪK2�����¶�973K��1173K����£�K1��K2��ֵ�ֱ����£�
�¶� | K1 | K2 |
973K | 1.47 | 2.38 |
1173K | 2.15 | 1.67 |
������������⣺
(1)ͨ�������е���ֵ�����ƶϣ���Ӧ����______��������������������������Ӧ��
(2)���з�Ӧ����CO2��g��+H2��g��![]() CO��g��+H2O��g��������д���÷�Ӧ��ƽ�ⳣ��K3�ı���ʽ��K3=_______��
CO��g��+H2O��g��������д���÷�Ӧ��ƽ�ⳣ��K3�ı���ʽ��K3=_______��
(3)����ͬ�¶��£����ݷ�Ӧ���������Ƶ���K3��K1��K2֮��Ĺ�ϵʽ____________���ݴ˹�ϵʽ���ϱ����ݣ�Ҳ���ƶϳ���Ӧ����_____��������������������������Ӧ��
(4)Ҫʹ��Ӧ����һ�������½�����ƽ��������Ӧ�����ƶ����ɲ�ȡ�Ĵ�ʩ��______ ����д��ĸ��ţ���ͬ����
A������ѹǿ B���������ʱ����ϡ������ C�������¶� D��ʹ�ú��ʵĴ��� E������ϵ��Ͷ������P2O5����
(5)��֪���Ŀ��淴ӦCO (g) ��H2O(g) ![]() CO2 (g) ��H2 (g) ��830Kʱ������ʼʱ��c(CO)��2mol/L��c(H2O)��3mol/L��ƽ��ʱCO��ת����Ϊ60%��ˮ������ת����Ϊ___________��K����ֵΪ_____________��830K����ֻ����ʼʱc(H2O)��Ϊ6mol/L����ˮ������ת����Ϊ_______��
CO2 (g) ��H2 (g) ��830Kʱ������ʼʱ��c(CO)��2mol/L��c(H2O)��3mol/L��ƽ��ʱCO��ת����Ϊ60%��ˮ������ת����Ϊ___________��K����ֵΪ_____________��830K����ֻ����ʼʱc(H2O)��Ϊ6mol/L����ˮ������ת����Ϊ_______��
����Ŀ���о����������̼��������ȴ�����Ⱦ��Ĵ���������Ҫ������
I. ��֪����N2(g)��O2(g)=2NO(g) ��H����180.5kJ��mol��1 ��CO��ȼ������H= - 283 kJ��mol��1
��1�������������ڰ�װ�Ĵ�ת��������ʹβ������Ҫ��Ⱦ�NO��CO��ת��Ϊ���Ĵ���ѭ�����ʣ��÷�Ӧ���Ȼ�ѧ����ʽΪ_________________________________________________��
��2��һ���¶��£��ں����ܱ������г���1 molNO��2 molCO���и÷�Ӧ���������жϷ�Ӧ�ﵽ��ѧƽ��״̬����_______��
�ٵ�λʱ��������a mo1 CO��ͬʱ����2a mol NO
�ڻ�������ƽ����Է����������ٸı�
�����������NO��CO�����ʵ���֮�ȱ��ֲ���
�����������ܶȲ��ٸı�
II. ��������������������Լ�����������ӦΪ2NO2(g)��O3(g)![]() N2O5(g)��O2(g)����ס������������Ϊ1.0 L�ĺ����ܱ������зֱ����2.0 mol NO2��1.0 mol O3���ֱ���T1��T2�¶��£�����һ��ʱ���ﵽƽ�⡣��Ӧ������n(O2)��ʱ�䣨t���仯������±���
N2O5(g)��O2(g)����ס������������Ϊ1.0 L�ĺ����ܱ������зֱ����2.0 mol NO2��1.0 mol O3���ֱ���T1��T2�¶��£�����һ��ʱ���ﵽƽ�⡣��Ӧ������n(O2)��ʱ�䣨t���仯������±���
t/s | 0 | 3 | 6 | 12 | 24 | 36 |
��������T1��n(O2)/mol | 0 | 0.36 | 0.60 | 0.80 | 0.80 | 0.80 |
��������T2��n(O2)/mol | 0 | 0.30 | 0.50 | 0.70 | 0.85 | 0.85 |
��3��T1___T2����>��<��=����ͬ�����÷�Ӧ��H___0 ����������________________________________��
��4���������У�0~3s�ڵ�ƽ����Ӧ����v(NO2)=______________________��
��5����������NO2ƽ��ת����Ϊ________��T1ʱ�÷�Ӧ��ƽ�ⳣ��Ϊ_______________________��
III. ��NO2(g)ת��ΪN2O4(g)������
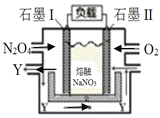
��6��ʯīIΪ______����ѡ������������Y��ѧʽΪ_________��
��7��II�缫��ӦʽΪ_________________________________________��

