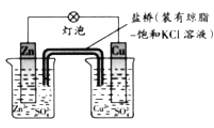
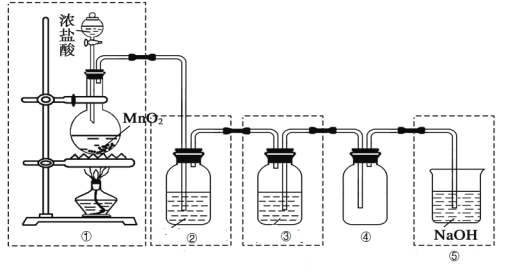
��Ŀ����
����Ŀ��CH4��������CO2�����ɵõ�����CO�Ļ���ԭ�ϡ��ش��������⣺
��1��CH4��������CO2�Ĵ�ת����ͼ��ʾ��

����֪��ط�Ӧ�������仯��ͼ��ʾ��

���̢���Ȼ�ѧ����ʽΪ________��
�ڹ����������̢��˵������ȷ����________������ţ���
a��ʵ���˺�̼�����뺬�����ʵķ���
b���ɱ�ʾΪCO2��H2��H2O��g����CO
c��COδ���뷴Ӧ
d��Fe3O4��CaOΪ�����������˷�Ӧ�Ħ�H
�������������䣬�ڲ�ͬ�������������£���ӦCH4��g����CO2��g����2CO��g����2H2��g��������ͬʱ���CH4��ת�����淴Ӧ�¶ȵı仯��ͼ��ʾ��a����������״̬________����ǡ����ǡ���ƽ��״̬��b��CH4��ת���ʸ���c�㣬ԭ����________��
��2����һ�����ܱ������У�CH4��CO2�ķ�ѹ�ֱ�Ϊ20kPa��25kPa������Ni������Al2O3������������1123Kʹ�䷢����ӦCH4��g����CO2��g����2CO��g����2H2��g����

���о�����CO���������ʦԣ�CO����1.3��10��2��p��CH4����p��CO2��mol��g��1��s��1��ijʱ�̲��p��CO����20kPa����p��CO2����________kPa���ԣ�CO����________mol��g��1��s��1��
�ڴﵽƽ�������ϵѹǿ����ʼʱ��1.8������÷�Ӧ��ƽ�ⳣ���ļ���ʽΪKp��________��kPa��2�����ø����ʵķ�ѹ�������ʵ���Ũ�ȼ��㣩
��3��CH4��������CO2�õ���CO��ż����Ӧ���Ƶò��ᣨH2C2O4���������£���ijŨ�ȵIJ�����Һ�м���һ��Ũ�ȵ�NaOH��Һ��������Һ��![]() �����ʱ��Һ��pH��________������֪������H2C2O4��Ka1��6��10��2��Ka2��6��10��5��lg6��0.8��
�����ʱ��Һ��pH��________������֪������H2C2O4��Ka1��6��10��2��Ka2��6��10��5��lg6��0.8��
���𰸡�CH4(g)+CO2(g)=2CO(g)+2H2(g) ��H=247.4kJ��mol-1 cd ���� b��c��δ��ƽ�⣬b���¶ȸߣ���Ӧ���ʿ죬��ͬʱ����ת���ʸ� 15 1.95 ![]() 2.7
2.7
��������
����������Ӧ�������ߵ��Ȼ�ѧ����ʽ��Ӧ�ø�˹���ɿɵù���I���Ȼ�ѧ����ʽ������II�н���ѭ���������������Ӧ���ѭ��������������������ɵ��ܷ�Ӧ����ʽ�����º���ʱ�������ķ�ѹ�������ʵ��������ȣ����÷�ѹ�������ʵ������м��㡣������Һ��NaOH��Һ��Ϻ�����������ƽ����Ȼ���ڣ��ݵ��볣������ʽ�����ض���������Һ��pH��
(1)����CH4��������CO2�Ĵ�ת��ͼ������I�Ļ�ѧ��ӦΪCH4(g)+CO2(g)=2CO(g)+2H2(g)������������Ӧ�������ߵ��Ȼ�ѧ����ʽ��
CH4(g)+H2O(g)=CO(g)+3H2(g) ��H��+206.2kJ��mol-1 ��i��
CO2(g)+4H2(g)=CH4(g)+2H2O(g) ��H����165kJ��mol-1 ��ii��
(i)��2+(ii)�ù���I���Ȼ�ѧ����ʽ��CH4(g)+CO2(g)=2CO(g)+2H2(g) ��H=247.4kJ��mol-1
�����������ʱ仯Ϊ�����ϣ�CO��H2��CO2��+ ���£��������壩�� ���£�H2O��+ ���ϣ�CO���������壩���ܷ�ӦΪH2+CO2=H2O+CO��Fe3O4��CaOΪ�ܷ�Ӧ�Ĵ������ܽ��ͷ�Ӧ�Ļ�ܣ������ܸı䷴Ӧ����H����ab��ȷ��cd����
��ͨ���������ܼӿ췴Ӧ���ʣ����̷�Ӧ����ƽ���ʱ�䡣����������ʹƽ�ⷢ���ƶ��������ܸı�ƽ��ת���ʡ���ͼ��a��Ϊ��ѧƽ�⣬���¶Ȳ��䣨800������������II����I��III��CH4ת����Ӧ���䣬��a�㲻�ǻ�ѧƽ�⡣
ͬ����ͼ��b��c���㶼δ�ﵽ��ѧƽ�⡣�����⣬b��c����ֻ���¶Ȳ�ͬ��b���¶Ƚϸߣ���Ӧ���ʿ죬��ͬʱ����CH4ת���ʸߡ�
(2)�پ���̬����PV��nRT�����º���ʱij�������ķ�ѹ�������ʵ��������ȡ���Ӧ�з�ѹΪ
1123K����ʱ��CH4��g����CO2��g����2CO��g����2H2��g��
��ʼ��ѹ/kPa�� 20 25 0 0
�ı��ѹ/kPa�� 10 10 20 20
ijʱ��ѹ/kPa�� 10 15 20 20
��ijʱ��p(CO2)��15kPa��p(CH4)��10kPa��������(CO)��1.3��10��2��p(CH4)��p(CO2)mol��g��1��s��1��1.95mol��g��1��s��1��
����ﵽƽ��ʱCH4�ĸı��ѹΪx kPa��
1123K����ʱ��CH4��g����CO2��g����2CO��g����2H2��g��
��ʼ��ѹ/kPa�� 20 25 0 0
�ı��ѹ/kPa�� x x 2x 2x
ƽ���ѹ/kPa�� 20��x 25��x 2x 2x
�����⣬��![]() ��1.8�����x��18��CH4(g)��CO2(g)��CO(g)��H2(g)��ƽ���ѹ������2 kPa��7 kPa��36 kPa��36 kPa������Kp��
��1.8�����x��18��CH4(g)��CO2(g)��CO(g)��H2(g)��ƽ���ѹ������2 kPa��7 kPa��36 kPa��36 kPa������Kp��![]() ��
��![]() (kPa)2��
(kPa)2��
(3)�����£�������Һ��NaOH��Һ��ϣ����û����Һ���Դ��ڷֲ����룺
H2C2O4![]() H++HC2O4�� Ka1��
H++HC2O4�� Ka1��![]()
HC2O4��![]() H++C2O42�� Ka2��
H++C2O42�� Ka2��![]()
��![]() ʱ��Ka1��Ka2��
ʱ��Ka1��Ka2��![]() ��
��![]() ��
��![]() ��6��10��3.5 mol/L��pH��2.7��
��6��10��3.5 mol/L��pH��2.7��

����Ŀ���±������ڱ��е�һ���֣����ݢ�--�������ڱ��е�λ�ã���Ԫ�ط�������ѧʽ�ش��������⣺
������ | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
1 | �� | |||||||
2 | �� | �� | �� | �� | ||||
3 | �� | �� | �� | �� | �� |
��1������Ԫ�أ���ѧ��������õ���______����ԭ����ǿ�ĵ�����______���������ư뵼����ϵ�Ԫ����______��
��2���ڢڡ��ۡ��ܡ��ݡ��ޡ��ߡ��ࡢ���У�ԭ�Ӱ뾶������______��
��3���ȽϢޡ��ߡ�����⻯����ȶ�����������______��
��4����![]() λ��Ԫ�����ڱ��еڶ����ڵڢ�A�塣��ͬ�����£����������ᷴӦ�ȵ�������ᷴӦ����______��������������������������д���������ᷴӦ�Ļ�ѧ����ʽ__________��
λ��Ԫ�����ڱ��еڶ����ڵڢ�A�塣��ͬ�����£����������ᷴӦ�ȵ�������ᷴӦ����______��������������������������д���������ᷴӦ�Ļ�ѧ����ʽ__________��