��Ŀ����
ɭ���ǹ��õı�����Դ���������ṩľ�ĺͶ����ָ���Ʒ�������ڱ���������ά����̬ƽ�ⷽ�����ż�Ϊ��Ҫ�����ã��Իش�
(1)ɭ��ͨ��ʲô;������CO2����ֹ����ЧӦ��
_______________________________________________________________��
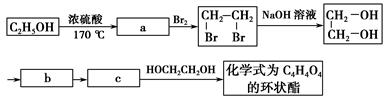
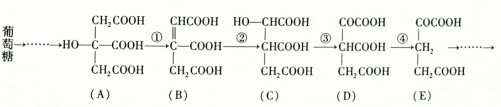
(2)ľ������Ҫ�Ļ���ԭ�ϣ�����Ҫ�ɷ�����ά�أ���ά�ط����ɺܶ��________��Ԫ���ɣ���ÿ����Ԫ�к���________���ǻ���������ά���ܷ���________��Ӧ������ŨH2SO4��ŨHNO3�Ļ�������Ӧ�Ļ�ѧ����ʽ��____________________________________��
(3)��ά����Ũ�����������ˮ������ղ�����________________(��ṹ��
ʽ)��д����ҽѧ�ϼ�������ʵĻ�ѧ����ʽ__________________��
(1)������á�(2)�����ǡ�3������
[C6H7O2(OH)3]n��3nHNO3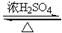 [C6H7O2(ONO2)3]n��3nH2O
[C6H7O2(ONO2)3]n��3nH2O
(3)CH2OH(CHOH)4CHO
CH2OH(CHOH)4CHO��2Cu(OH)2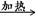 CH2OH(CHOH)4COOH��Cu2O����2H2O
CH2OH(CHOH)4COOH��Cu2O����2H2O
����

��ע���������վ�ڵı�������ijЩʡ�м���վ�ṩ�����ͱ����90#��93#������E90��E93���������������м�����10%��ȼ���Ҵ����Ƶõ��Ҵ����͡�ȼ���Ҵ����������̿�����ͼ��ʾ��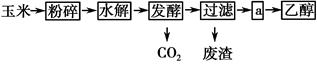
(1)��������Ŀ����_______________________________________________��
(2)����������Ϊ�˼������ˮ���Ƿ���ȫ����ʹ�õ��Լ���________��
(3)����a�IJ�����________(����ĸ)��
| A������ | B����ȡ | C������ | D����Һ |
(5)��������Ϊԭ�������Ҵ��Ļ�ѧ��Ӧ������ʽ��ʾ��
(C6H10O5)n��nH2O
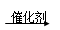 nC6H12O6
nC6H12O6C6H12O6�D��2C2H5OH��2CO2��
����������Ӧʽ���������100 kg���������Ͽ�������ˮ�Ҵ�________ kg��
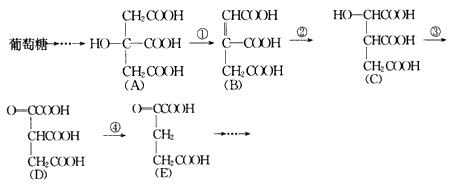

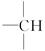 ��������Ȼ������ˮ������ղ���֮һ�����ͬ���칹��Ľṹ��ʽΪ_____________________________________��
��������Ȼ������ˮ������ղ���֮һ�����ͬ���칹��Ľṹ��ʽΪ_____________________________________��