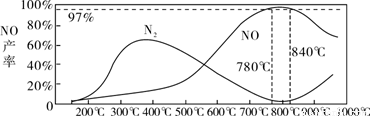
��Ŀ����
��ҵ�ϳɰ����Ʊ�����һ��������������������£�
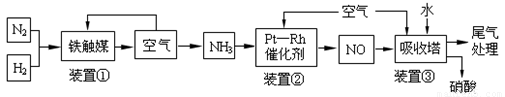
��ش��������⣺
��1��ij����С���о����������������������£��ı���ʼ�����������ʵ������Է�ӦN2(g)��3H2(g)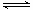 2NH3(g)
��H����92.4
kJ��mol-1��Ӱ�졣ʵ������ͼ��ʾ����ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ�����
2NH3(g)
��H����92.4
kJ��mol-1��Ӱ�졣ʵ������ͼ��ʾ����ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ�����
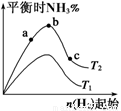
��ͼ����T2��T1�Ĺ�ϵ�ǣ�T2 T1(�>������<������=������ȷ����)��
�ڱȽ���a��b��c����������ƽ��״̬�У�N2��ת������ߵ��� (����ĸ)��
��Ҫʹ��Ӧ�İٷֺ������������ʼ��ϵ��ԭ��Ͷ�ϱ�n(H2)/n(N2) 3���� ��>������<������=������ȷ���������������ݻ���Ϊ1 L����ʼ״̬n(H2)=3 mol����Ӧ�ﵽƽ��ʱH2��ת����Ϊ60%�����������(T2)����Ӧ��ƽ�ⳣ��K= �����������С�������λ��
��2����֪��N2(g)��O2(g)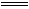 2NO(g) ��H����180.5 kJ��mol-1
2NO(g) ��H����180.5 kJ��mol-1
2H2(g)��O2(g)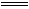 2H2O(g) ��H����483.6 kJ��mol-1
2H2O(g) ��H����483.6 kJ��mol-1
����17 g�����������侭��������ȫ��Ӧ������һ�����������ˮ��������ù��������ų�������Ϊ kJ��
��3����װ�â��У�NH3��O2��145��Ϳ�ʼ���з�Ӧ���ڲ�ͬ�¶Ⱥʹ������������ɲ�ͬ�������ͼ��ʾ����
�¶Ƚϵ�ʱ���� Ϊ�����¶ȸ���900��ʱ��NO�����½��Ŀ���ԭ���� ��
��14�֣�
��1���� ������ c �� �� ������2�֣���6�֣���2.08��2�֣�
��2��226.25kJ��226.3 kJ��3�֣�
��3��N2 ��1�֣���������������NO�ķ�ӦΪ���ȷ�Ӧ�������¶�ƽ�������ƶ���ת�����½���2�֣�
��������
���������
��1���ٺϳɰ���Ӧ�Ƿ��ȷ�Ӧ���¶�Խ�ߣ����İٷֺ���ԽС������T2<T1������������Ũ�ȣ�����ת������ߣ����Ե���ת������ߵ���c����Ҫʹ��Ӧ�İٷֺ�������������������ӦΪ1��3����2������������ӦΪ4NH3+5O2=4NO+6H2O���ɸ�˹���ɵ�4NH3(g)+5O2(g)=4NO(g)+6H2O(g) ��H����180.5��2+92.4��2��483.6��3��kJ��mol-1=��905 kJ��mol-1������1molNH3����������226.25kJ��
��3����ͼ���֪�¶ȵ�ʱ��Ҫ���ɵ��������Ĵ��������ȣ��¶ȸ���900��ʱ��ƽ�������ƶ���NO���ʽ��͡�
���㣺 ��ɳ����ԭ�� ��˹����

��1����ҵ����ʱ����ȡ������һ����ӦΪ��CO��g��+H2O��g��?CO2��g��+H2��g����t��ʱ����10L�ܱ������г���2mol CO��3molˮ��������Ӧ����ƽ�����ϵ��c��H2��=0.12mol?L-1������¶��´˷�Ӧ��ƽ�ⳣ��K=
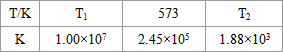
��2���ϳ����з�����ӦN2��g��+3H2��g��?2NH3��g����H��0���±�Ϊ��ͬ�¶��¸÷�Ӧ��ƽ�ⳣ�����ɴ˿���֪������T1
| T/�� | T1 | 300 | T2 |
| K | 1.00��107 | 2.45��105 | 1.88��103 |
��4���ð������������������ᣬ��β���е�NOx����Ⱦ������Ŀǰ��ѧ��̽������ȼ�������еļ���Ƚ����������ﻹԭΪ������ˮ����Ӧ����Ϊ��
CH4��g��+4NO2��g���T4NO��g��+CO2��g��+2H2O��g����H=-574kJ?mol-1
CH4��g��+4NO��g���T2N2��g��+CO2��g��+2H2O��g����H=-1160kJ?mol-1
�����ֱ�ӽ�NO2��ԭΪN2���Ȼ�ѧ����ʽΪ
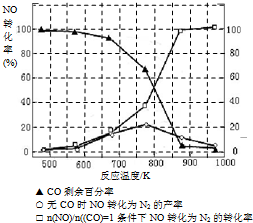
��5��ij�о�С����ʵ�����ԡ�Ag-ZSM-5��Ϊ��������ý�NOת��ΪN2��ת�������¶ȱ仯�������ͼ����ͼ����������ʹ��CO���¶ȳ���775�棬����NO��ת���ʽ��ͣ�����ܵ�ԭ��Ϊ
| n(NO) |
| n(CO) |

 ��3��NH3��O2�ڲ�ϵ���������´�145��Ϳ�ʼ��Ӧ��4NH3��g��+5O2��g��?4NO��g��+6H2O��g����H=-905kJ?mol-1����ͬ�¶���NO��������ͼ��ʾ���¶ȸ���900��ʱ��NO�����½���ԭ��
��3��NH3��O2�ڲ�ϵ���������´�145��Ϳ�ʼ��Ӧ��4NH3��g��+5O2��g��?4NO��g��+6H2O��g����H=-905kJ?mol-1����ͬ�¶���NO��������ͼ��ʾ���¶ȸ���900��ʱ��NO�����½���ԭ��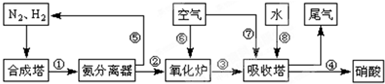
 ��3��N2��H2���������������´�145��Ϳ�ʼ��Ӧ����ͬ�¶���NH3������ͼ��ʾ���¶ȸ���900��ʱ��NH3�����½���ԭ��
��3��N2��H2���������������´�145��Ϳ�ʼ��Ӧ����ͬ�¶���NH3������ͼ��ʾ���¶ȸ���900��ʱ��NH3�����½���ԭ��


