��Ŀ����
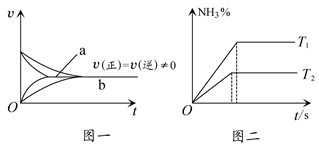
���ܱ������У���1.0 mol CO��1.0 mol H2O��ϼ��ȵ�800 �棬�������з�Ӧ��
CO(g)��H2O(g)  CO2(g)��H2(g)��һ��ʱ���÷�Ӧ�ﵽƽ�⣬���CO�����ʵ���Ϊ0.5 mol��������˵����ȷ���ǣ� ��
CO2(g)��H2(g)��һ��ʱ���÷�Ӧ�ﵽƽ�⣬���CO�����ʵ���Ϊ0.5 mol��������˵����ȷ���ǣ� ��
| A��800 ���£��÷�Ӧ�Ļ�ѧƽ�ⳣ��Ϊ0.25 |
| B��427 ��ʱ�÷�Ӧ��ƽ�ⳣ��Ϊ9.4����÷�Ӧ�Ħ�H��0 |
| C��800 ���£����������ƽ����ϵ��ͨ��1.0 mol��CO(g)����ƽ��ʱCO���ʵ�������Ϊ33.3% |
| D��800 ���£����������ƽ����ϵ��ͨ��1.0 mol��H2O(g) ����ƽ��ʱCOת����Ϊ66.7% |
D
����

��ϰ��ϵ�д�
 ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�
�����Ŀ
���з�Ӧ�У�ͬʱ����ͼ���͢����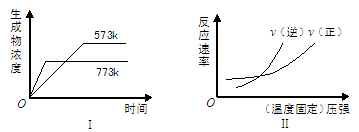
A��N2(g)��3H2(g)  2NH3(g) ��H��0 2NH3(g) ��H��0 |
B��2SO3(g)  2SO2(g)��O2(g) ��H��0 2SO2(g)��O2(g) ��H��0 |
C��H2(g)��CO(g) 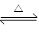 C(s)��H2O(g) ��H��0 C(s)��H2O(g) ��H��0 |
D��4NH3(g)��5O2(g) 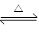 4NO(g)��6H2O(g) ��H��0 4NO(g)��6H2O(g) ��H��0 |
������NH4I�����ܱ������У���һ���¶��·������з�Ӧ��
��NH4I(s)  NH3(g) + HI(g) ��2HI(g)
NH3(g) + HI(g) ��2HI(g)  H2(g) + I2(g)
H2(g) + I2(g)
�ﵽƽ��ʱ��c��H2��=0.5mol/L��c��NH3��=4mol/L������¶��·�Ӧ�ٵ�ƽ�ⳣ��Ϊ
| A��9 | B��12 | C��16 | D��20 |
��ij���ݵ��ܱ������У����淴ӦA(g)��B(g)  xC(g)����ͼ��ʾ�Ĺ�ϵ���ߣ�����˵����ȷ���ǣ� ��
xC(g)����ͼ��ʾ�Ĺ�ϵ���ߣ�����˵����ȷ���ǣ� ��
| A���¶ȣ�T1��T2 | B��ѹǿ��p1��p2 |
| C������Ӧ�����ȷ�Ӧ | D��x��ֵ��2 |
��2NO2 N2O4�Ŀ��淴Ӧ��,����״̬һ������ƽ��״̬����(����)
N2O4�Ŀ��淴Ӧ��,����״̬һ������ƽ��״̬����(����)
| A��N2O4��NO2�ķ�������Ϊ1��2 |
| B��N2O4��NO2��Ũ����� |
| C��ƽ����ϵ����ɫ���ٸı� |
| D����λʱ������1 mol N2O4��ΪNO2��ͬʱ,��2 mol NO2���� |
N2+3H2 2NH3��Ӧ�ﵽƽ��ʱ��˵������ȷ����(����)��
2NH3��Ӧ�ﵽƽ��ʱ��˵������ȷ����(����)��
| A��ƽ����ϵ�е������������������ʵ������ֲ��� |
| B��ƽ����ϵ�е���������������Ũ�ȱ��ֲ��� |
| C����λʱ�������ɵİ��ͷֽ�İ����ʵ�����ͬ |
| D����λʱ�������ĵĵ��������������ɵİ������ʵ�����ͬ |
 2A(g) �� B(g)�÷�Ӧ��ƽ�ⳣ������ʽΪK=c(A)2��c(B)
2A(g) �� B(g)�÷�Ӧ��ƽ�ⳣ������ʽΪK=c(A)2��c(B)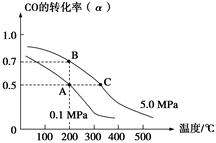
 2NH3(g)����H<0���ٶ�Ϊv���������¶ȣ�����Ӧ���ʼӴ�
2NH3(g)����H<0���ٶ�Ϊv���������¶ȣ�����Ӧ���ʼӴ�