��Ŀ����
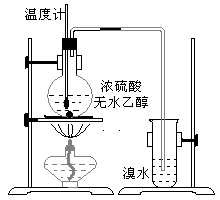
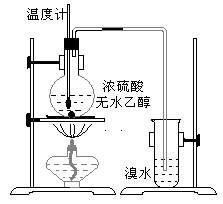
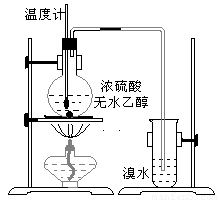
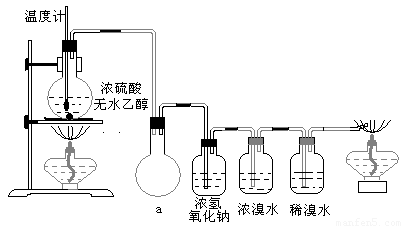
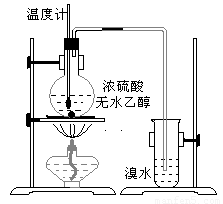
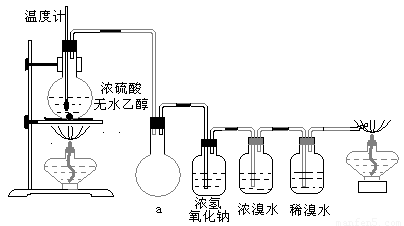
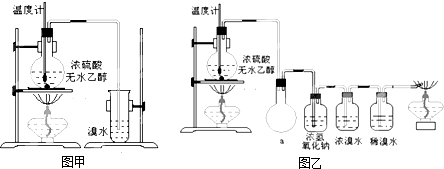
Ϊ̽��ʵ��������ϩ����ϩ����ˮ�ļӳɷ�Ӧ����ͬѧ�������ͼ����ʾ��ʵ��װ�ã���������ʵ�飮���¶�����170������ʱ���д����������������������ͨ����ˮ�У���ˮ����ɫѸ����ȥ����ͬѧ��Ϊ�ﵽ��ʵ��Ŀ�ģ���ͬѧ��ϸ�۲��˼�ͬѧ������ʵ����̣����ֵ��¶�����100������ʱ����ɫҺ�忪ʼ��ɫ����160������ʱ�����Һȫ�ʺ�ɫ����170�泬�������������ٶ����Լӿ죬���ɵ������д̼�����ζ���ɴ����Ƴ���������������Ӧ�����ʣ�����Ӱ����ϩ�ļ���������ȥ��
�ݴ˻ش��������⣺
��1��д����ͬѧʵ������Ϊ�ﵽʵ��Ŀ��������Ӧ�Ļ�ѧ����ʽ��
��2����ͬѧ�۲쵽�ĺ�ɫ������
��3����ͬѧ���ݼ���ͬѧ�ķ�������Ϊ��������CO��CO2�������������Ϊ֤��CO���ڣ����������ͼ�ҹ��̣��ù��̿ɰ�ʵ���в������л����������������������徭��ȼ����ɫ���棬ȷ����һ����̼��
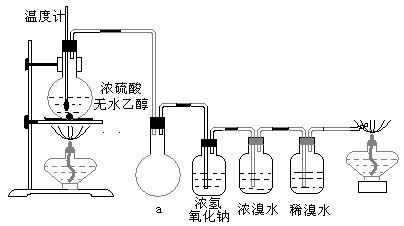
�����װ��a��������
��Ũ��ˮ��������

�ݴ˻ش��������⣺
��1��д����ͬѧʵ������Ϊ�ﵽʵ��Ŀ��������Ӧ�Ļ�ѧ����ʽ��
CH3CH2OH
CH2=CH2��+H2O
| Ũ���� |
| 170�� |
CH3CH2OH
CH2=CH2��+H2O
��| Ũ���� |
| 170�� |
CH2=CH2+Br2��CH2BrCH2Br
CH2=CH2+Br2��CH2BrCH2Br
����2����ͬѧ�۲쵽�ĺ�ɫ������
C
C
���̼���������SO2
SO2
����ͬѧ��Ϊ�̼�������Ĵ��ھͲ�����Ϊ��ˮ��ɫ����ϩ�ļӳɷ�Ӧ��ɵģ�ԭ���ǣ��û�ѧ���̱�ʾ����SO2+Br2+2H2O=H2SO4 +2HBr
SO2+Br2+2H2O=H2SO4 +2HBr
����3����ͬѧ���ݼ���ͬѧ�ķ�������Ϊ��������CO��CO2�������������Ϊ֤��CO���ڣ����������ͼ�ҹ��̣��ù��̿ɰ�ʵ���в������л����������������������徭��ȼ����ɫ���棬ȷ����һ����̼��
�����װ��a��������
��ȫװ�ã�������
��ȫװ�ã�������
��Ũ��ˮ��������
������ϩ��������������
������ϩ��������������
��ϡ��ˮ��������������ϩ�����������Ƿ����
������ϩ�����������Ƿ����
����������1��ʵ�����Ʊ���ϩ���õ�ԭ��Ϊ�Ҵ���Ũ��������������ˮ������Ӧ�����Ǽ��ȵ�170�棻��ˮ�е�������ϩ�����ӳɷ�Ӧ��
��2��Ũ�������ǿ�����ԣ��������Ҵ�������������巢��������ԭ��Ӧ��
��3������ɵ�����������װ���ڵĴ���ѹ������Ĵ���ѹ��һ���IJ�ֵ��
��Ũ��ˮ�ܺ���ϩ�Ͷ�������Ӧ��
��2��Ũ�������ǿ�����ԣ��������Ҵ�������������巢��������ԭ��Ӧ��
��3������ɵ�����������װ���ڵĴ���ѹ������Ĵ���ѹ��һ���IJ�ֵ��
��Ũ��ˮ�ܺ���ϩ�Ͷ�������Ӧ��
����⣺��1�������Ҵ���Ũ����Ĵ������·�����������ˮ��ȡ��ϩ���Ҵ���������ȥ��Ӧ��CH3CH2OH
CH2=CH2��+H2O����ˮ�е�������ϩ�����ӳɷ�Ӧ����CH2=CH2+Br2��CH2BrCH2Br���ʴ�Ϊ��CH3CH2OH
CH2=CH2��+H2O��CH2=CH2+Br2��CH2BrCH2Br��
��2����ͬѧ��ϸ�۲��˼�ͬѧ������ʵ����̣����ֵ��¶�����100������ʱ����ɫҺ�忪ʼ��ɫ����160������ʱ�����Һȫ�ʺ�ɫ��Ũ�������ǿ�����ԣ��������Ҵ������Ҵ�������̼��ͬʱ��������ԭ�ɶ���������������巢��������ԭ��Ӧ��������������SO2+Br2+2H2O=H2SO4 +2HBr���ʴ�Ϊ��C��SO2��SO2+Br2+2H2O=H2SO4 +2HBr��
��3������ϩ��������ˮ�У�װ���ڵĴ���ѹ������Ĵ���ѹ��һ���IJ�ֵ�����װ��a�ɷ�ֹŨ��ˮ����������ϩ�ķ�Ӧ�����У��ʴ�Ϊ����ȫװ�ã���������
����ˮ�����������������ԭ��Ӧ���屻����������+4�۵���ԭ��-1�۵��壬+4�۵�����������+6�۵�����ˮ��ɫ����ϩ����ˮ�е��巢���ӳɷ�Ӧ����ˮ��ɫ���ɼ�����ϩ�Ĵ��ڣ�����������巢��������ԭ��Ӧ��������������ᣬ�ɼ����������Ĵ��ڣ��ʴ�Ϊ��������ϩ�������������壻������ϩ�����������Ƿ������
| Ũ���� |
| 170�� |
| Ũ���� |
| 170�� |
��2����ͬѧ��ϸ�۲��˼�ͬѧ������ʵ����̣����ֵ��¶�����100������ʱ����ɫҺ�忪ʼ��ɫ����160������ʱ�����Һȫ�ʺ�ɫ��Ũ�������ǿ�����ԣ��������Ҵ������Ҵ�������̼��ͬʱ��������ԭ�ɶ���������������巢��������ԭ��Ӧ��������������SO2+Br2+2H2O=H2SO4 +2HBr���ʴ�Ϊ��C��SO2��SO2+Br2+2H2O=H2SO4 +2HBr��
��3������ϩ��������ˮ�У�װ���ڵĴ���ѹ������Ĵ���ѹ��һ���IJ�ֵ�����װ��a�ɷ�ֹŨ��ˮ����������ϩ�ķ�Ӧ�����У��ʴ�Ϊ����ȫװ�ã���������
����ˮ�����������������ԭ��Ӧ���屻����������+4�۵���ԭ��-1�۵��壬+4�۵�����������+6�۵�����ˮ��ɫ����ϩ����ˮ�е��巢���ӳɷ�Ӧ����ˮ��ɫ���ɼ�����ϩ�Ĵ��ڣ�����������巢��������ԭ��Ӧ��������������ᣬ�ɼ����������Ĵ��ڣ��ʴ�Ϊ��������ϩ�������������壻������ϩ�����������Ƿ������
���������⿼������ϩ��ʵ�����Ʒ��Լ�����ļ��飬����������ʵĻ�����ѧ���ʣ��ǽ����Ĺؼ���ƽʱ��ע�������ط�Ӧ֪ʶ���Ѷ��еȣ�

��ϰ��ϵ�д�
 ���źþ���Ԫ����ĩ��ϵ�д�
���źþ���Ԫ����ĩ��ϵ�д�
�����Ŀ