��Ŀ����
ij��ѧ��ȤС��ͬѧ���̡�֤�������д��ڵ�Ԫ�ء���ʵ��ʱ���Կα��ϵ����������������պ�Ļҽ��е�Ԫ����I-��ʽ���ڡ����������ʡ�����KI���屻�������ʣ���dz��ɫ����I2����KI����ֱ�Ӽ���ʱ��Ϊʲô���������أ���ˣ�С��ͬѧ���I-������������������֮���Ҫʲô����������ʵ��̽����
��������衿���ݾ���KI����ᱻ���������ʣ���Ͽ����ijɷ֣������ų� ��ϡ�������Ӱ�졣Ȼ���KI������������������裺
����һ����ҪH2O�IJ��룻
���������ҪCO2�IJ��룻
�������� ��
��ʵ����֤��
(1)Ϊ����֤�������С��ͬѧ���������ʵ�鷽����ȡ10mLKI��Һ(Ũ�Ƚϴ�)��5֧�Թ��У����Թ���ͨ��CO2������ߵμ����ᡣһ��ʱ���Ա��Թ��е���ɫ��dz��֮�����Թ��е��������Һ���ٴζԱ��Թ��е���ɫ��dz������ʵ��������±���
| �Թ���� | 1 | 2 | 3 | 4 | 5 |
| ͨ��CO2ʱ��/min | 0 | 1 | 5 | | |
| �����/�� | 0 | | | 3 | 6 |
| ��Һ����ɫ�Ա� | ��ɫ | dz��ɫ����ɫ�����μ�� | |||
| �μӵ�����Һ�����ɫ�Ա� | ��ɫ | dz��ɫ����ɫ�����μ�� | |||
С��ͬѧͨ��ʵ����������˼����CO2�ڷ�Ӧ�����е����ã��ó����ۣ�CO2����ͬ����һ����KI�������Ĺ������ṩ ������
д����ʵ����CO2��KI��Ӧ�Ļ�ѧ����ʽ ��
(2)Ϊ����֤I-�������Ƿ����Ҫ��H2O�IJ��룬С��ͬѧ���������ʵ����֤��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ��ȡһ�ݴ�����KI���壬�ֳ����ȷݣ� | |
| ��һ�ݼ��뵽װ�� �ļ���ƿA�У� һ�ݼ��뵽װ�� �ļ���ƿA�У� �ۼ����۲졣 | |
��������ߡ�
���ڿ�����CO2��ˮ�����ĺ����ܵͣ������ڼ���KI�Ĺ�������������������ױ�����ɢ�������ܺܺõ���KI�Ӵ���KI��Ȼ�Ͳ��ܱ���������ʱ�䱣��⻯����Ҫ�ܹ⡢ ��
��13�֣�
[�������] N2��1�֣� �� CO2��H2O�Ĺ�ͬ���루2�֣���
[ʵ����֤] ��1�����ԣ�1�֣���4KI + O2 + 2CO2 ="=" 2K2CO3 + 2I2��3�֣�
��2��ʵ�鲽�裺 Ԥ������ͽ��� ��ȡһ�ݴ�����KI���壬�ֳ����ȷݣ� �ڸ����CO2��O2������壨1�֣�
��ʪ��CO2��O2������壨1�֣�������ƿA�е�KI������ɫ��ƣ�������KI���Բ���ҪH2O�μӣ�1�֣���
������ƿA�е�KI������ɫ���䣬������ƿB��KI������ɫ��ƣ�������KI����Ҫ��H2O�μӣ�1�֣���
�������������֣�
[�������] �ܷ��ҷ����ڸ��ﴦ��2�֣�
�����������������ı����Ƕ�����������̽����������Ӧ������Ϊ�����Ӻ�������������H2O��CO2�е�һ�ֻ����֣���̽���ķ������Χ��H2O��CO2��H2CO3���Ӷ����ʵ�����֤������ʵ������п������Ա�ʵ��Ͳ���ʵ�顣��ʵ����ƹ����У�����Ҫ�Ա�����H2O���뷴Ӧ��Ӧ������CO2��O2��CO2��O2��H2O�����鷴Ӧ���жԱȴӶ��õ����ۡ�
�ڽ��������Ҫע���������������α�֪ʶ�Ľ�ϣ����Խ����������Ļ������̽��н����������Ҫ���롢�������������������ȷ���淶��
���㣺������̽��ʵ��Ϊ����������Ԫ�ؼ�������֪ʶ��ʵ��̽���������������֪ʶ��

 ��ĩ�����ϵ�д�
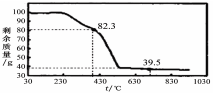
��ĩ�����ϵ�д�CuSO4��Һ��Na2CO3��Һ��ϲ�������ɫ������������ij��ȤС��Գ�����ɵ�̽����
��������衿
����1������ΪCu (OH)2
����2������Ϊ
����3������Ϊ��ʽ̼��ͭ[��ѧʽ�ɱ�ʾΪnCuCO3��mCu (OH)2]
���������ϡ���������һ�ֳ������Ⱦ��ֽ⣨����������ᾧˮ����
������̽����
����1������������Һ���ˣ�������ˮϴ�ӣ�������ˮ�Ҵ�ϴ�ӣ���ɣ�
����2����ͬѧȡһ�������壬�����������õ�����װ�ã��г�����δ���������ж���ʵ�飻
��1������Ӧ��A������ɫ�����ڣ�C������������֤������ ������
��2����ͬѧ��ΪֻҪ����ͼ��Bװ�õ��Լ���������ij�Լ������֤�������м��裬���Լ��� ������ţ���
a��Ũ���� b����ˮCuSO4 c����ʯ�� d��P2O5
��3����ͬѧ����B�Լ�����֤����3������ʵ�������� ��
������̽����
��4����ͬѧ��һ��̽������3�й������ɣ�
����ͬѧ���һЩ������20������ݣ����±�����C�еij���ʯ��ˮ��ΪBa(OH)2��Һ����ԭ���� ��˫ѡ������ţ�
| �ܽ��(S)/g | �ܶȻ�(Ksp) | Ħ������(M)/g��mol��1 | |||
| Ca(OH)2 | Ba(OH)2 | CaCO3 | BaCO3 | CaCO3 | BaCO3 |
| 0.16 | 3.89 | 2.9��10-9 | 2.6��10-9 | 100 | 197 |
b��Ba(OH)2Ϊǿ�Ca(OH)2Ϊ����
c�����յ���CO2���ɵ�BaCO3����������CaCO3���������С
d����ͬ�����£�CaCO3���ܽ�����Դ���BaCO3
������ȡ����ɫ��������Ϊ54.2 g��ʵ�������װ��B����������5.4 g��C�еIJ�������������Ϊ39.4 g���������ɫ����Ļ�ѧʽΪ ��
S2Cl2�ǹ�ҵ�ϳ��õ�����ʵ�����Ʊ�S2Cl2�ķ�����2�֣�
�� CS2+3Cl2 CCl4+S2Cl2���� 2S+Cl2
CCl4+S2Cl2���� 2S+Cl2 S2Cl2��
S2Cl2��
��֪S2Cl2����Ԫ����+1�ۣ�����ʽ�� �������ȶ�����ˮ������᪻���Ӧ��һ������Ԫ�ؼ�̬���ߣ�һ���ֽ��ͣ�����Ӧ�漰�ļ������ʵ��۷е����£�
�������ȶ�����ˮ������᪻���Ӧ��һ������Ԫ�ؼ�̬���ߣ�һ���ֽ��ͣ�����Ӧ�漰�ļ������ʵ��۷е����£�
| ���� | S | CS2 | CCl4 | S2Cl2 |
| �е�/�� | 445 | 47 | 77 | 137 |
| �۵�/�� | 113 | -109 | -23 | -77 |
ʵ������������װ���Ʊ�S2Cl2�����ּг���������ȥ����

�ش��������⣺
��1��װ��B��C�в������������ƣ� ����Ӧԭ������д������ţ��� ��
��2��ʵ���������Լ�ͨ������36.5%��Ũ��Һ������ϡ����������� ��
��3��D������������������˫�����ã�����ȴˮ��������������������������ͬ����ͼ����������ȴ��ʽ��Ӧ�������и��л�ѧ�� ʵ�顣
A��ʯ�ͷ��� B����ȡ�屽 C����ȡ�������� D���Ʊ���˾ƥ��
��4��Bװ����ʢ�ŵ��� ����Ӧ���������ƿ�ڻ�����з������Ʒ�ķ����� ��D�в�����ˮԡ���ȵ�ԭ���� ��
��5��A��������װ��ʱ���ź�����̨��Ӧ�ȹ̶� �����������ƣ�������װ��װ����Ϻ�Ӧ�Ƚ��� �������Լ���ʵ����ϣ�A�в��ٲ�������ʱ���ɲ��װ�á����ʱ�����ȵIJ���Ӧ���� ��
��6��ʵ������У���ȱ��Cװ�ã����ֲ�Ʒ���Dz��壬���ָ������ԭ����û�ѧ����ʽ��ʾΪ ��ʵ����ϣ�����ʣ��Ũ���ᵹ��E�ձ�����������β��������������Һ���ʱ����������������ɫ�̼�����������������������ԭ���ǣ� �������ӷ���ʽ��ʾ����
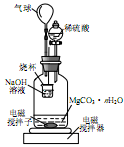
ijУ��ѧС���ͬѧ������̼��Ϊ�缫����Ȼ�ͭ��Һʱ��������̼���ϳ����к�ɫ���������⣬����������ɫ����������Ϊ̽������̼���ϵIJ����������¹��̣�
�������
ͭ�Ļ�������ɫ�������£�
| ���� | ��ɫ������ | ���� | ��ɫ������ |
| ������ͭCu(OH)2 | ��ɫ���岻����ˮ | ����ͭ��CuSO4�� | ��Һ����ɫ |
| ������ͭ��CuO�� | ��ɫ���岻����ˮ | �Ȼ�ͭ��CuCl2�� | ��Һ����ɫ��ϡ��Һ����ɫ |
| �Ȼ���ͭ��CuCl�� | ��ɫ���岻����ˮ | ��ʽ�Ȼ�ͭ | ��ɫ���岻����ˮ |
��1��������裺�ٺ�ɫ����һ����ͭ����������Cu2O���ڰ�ɫ����Ϊͭ�Ļ�����仯ѧʽ����ΪCuCl
��2��ʵ����֤��ȡ���CuCl2��Һ�������̼����ϴ�ӡ������������װ�ý���ʵ�飬��֤��������

ʵ�������̼���ϵİ�ɫ���ʱ�Ϊ��ɫ����ˮ����ͭ����ɫ��
��̼���ϵĺ�ɫ�����Ƿ���Cu2O ����ǡ�����������
������cװ�ú�����ͨ�� �У������ְ�ɫ��������˵�����������еİ�ɫ����һ�����ڣ���д��װ��b�з�����Ӧ�Ļ�ѧ����ʽ��
��
��3���������ۣ�
�ٵ��CuCl2��Һ��������Ϸ����ķ�ӦΪ��Cu2++2e-��Cu�� ������ʯī�缫��ⱥ������ͭ��Һ���۲�����̼��������ֵ������ɫ���ʸ��ţ��ް�ɫ���ʣ����� �������⣬���ְ�ɫ���ʡ�
Ư����һ�ֳ��õ���������
��1����ҵ������Ư�۷�Ӧ�Ļ�ѧ����ʽΪ��________________ __��Ư�۵���Ч�ɷ�Ϊ ��
��2��ij̽��С����г��Ϲ�����һ����װ�����Ư�ۣ��Ը�Ư�۵ijɷֽ���̽�������������Լ������ʵ�鷽��������ʵ�顣���ڴ�������ʵ�鱨�档
��ѡ�Լ���2mol��L��1NaOH��Һ��2mol��L��1HCl��Һ��2mol��L��1HNO3��Һ��0.5mol��L��1BaCl2��Һ��0.01mol��L��1AgNO3��Һ������ʯ��ˮ��ʯ����Һ����̪��Һ������ˮ��
| ʵ�鲽�� | Ԥ����������� |
| ����1��ȡ����Ư��������������ˮ����ֽ��裬���ã����ˣ��ó�������Һ�� | |
| ����2���������������2mol��L��1HCl��Һ��������������ͨ�� | ���� ���ۣ� |
| ����3��ȡ��Һ��װA��B��֧�Թܡ���A�Թܣ� | ������Һ�ȱ��ɫ��Ȼ����ɫ�� ���ۣ� |
| ����4����B�Թܣ� | ��������ɫ������ ���ۣ� |
��3��̽��С��Ϊ�ⶨƯ����Ca(ClO)2�ĺ�������ȡƯ��bg��ˮ�ܽ�����Ƴ�100mL��Һ��ȷ��ȡ25.00mL����ƿ���������������KI��Һ����ַ�Ӧ����Һ�е��������0.1000mol/L��Na2S2O3��Һ�ζ����ζ�2�Σ�ƽ������Na2S2O3��Һ20.00mL�����Ư����Ca(ClO)2����������Ϊ_____________ _����ֻ����ʽ���������㣬��֪��Mr[Ca(ClO)2]="143" ��Ca(ClO)2+4HCl=2Cl2��+CaCl2+2H2O��2Na2S2O3+I2=Na2S4O6+2NaI��
ʵ�����Ʊ����ռ������SO2�������������¡�װ�ã�����SO2���������������Ӹ�������˳��Ϊ��ӣ� ���ӣ� ���ӣ� ���ӣ� ���ӣ棨��ӿ���ĸ�� �� ��
| A��b c d e | B��d e b c | C��d e c b | D��e d b c |




 ������ƿ�У���0.05mol/L������Һ���еζ������ı���Һ20.00mL����ش��������⣺
������ƿ�У���0.05mol/L������Һ���еζ������ı���Һ20.00mL����ش��������⣺