��Ŀ����
��Ȫ��һ�ֳ�������Ȼ�����������ԭ���Ǵ���ѹǿ���ȡ�����������Ȫʵ�飨ͼ�мг�װ�þ�����ȥ����
��1��д��ʵ������ȡ�����Ļ�ѧ����ʽ�� _________ ��
��2���ռ�����Ӧʹ�� ����
��3��Ҫ�õ�����İ�����ѡ������ ���������
��4����ͼ1װ�ý�����Ȫʵ�飬��ƿ��װ�����ﰱ��������ˮ����IJ����� ����ʵ���ԭ���� ��
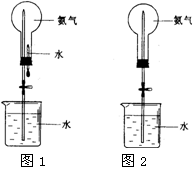
��5�����ֻ�ṩ��ͼ2��װ�ã������˵��������Ȫ�� ��
��1��д��ʵ������ȡ�����Ļ�ѧ����ʽ�� _________ ��
��2���ռ�����Ӧʹ�� ����
��3��Ҫ�õ�����İ�����ѡ������ ���������
| A��Ũ���� | B����ʯ�� | C��NaOH���� | D��P2O5���� |

��5�����ֻ�ṩ��ͼ2��װ�ã������˵��������Ȫ�� ��
��1��2NH4Cl+Ca((OH)2 CaCl2+2NH3��+2H2O��2�������ſ�������3��BC��4������ͷ�ι��е�ˮ������ƿ�У��ɿ����ɼУ�������ˮ�ܽ�����ƿ�е�NH3��ʹƿ��ѹǿ��С����ƿ�����γ�����ѹ�5���ñ�ë����ס��ƿһ��ʱ�䣬���ɿ����ɼС�������������Ҳ�ɣ�
CaCl2+2NH3��+2H2O��2�������ſ�������3��BC��4������ͷ�ι��е�ˮ������ƿ�У��ɿ����ɼУ�������ˮ�ܽ�����ƿ�е�NH3��ʹƿ��ѹǿ��С����ƿ�����γ�����ѹ�5���ñ�ë����ס��ƿһ��ʱ�䣬���ɿ����ɼС�������������Ҳ�ɣ�
 CaCl2+2NH3��+2H2O��2�������ſ�������3��BC��4������ͷ�ι��е�ˮ������ƿ�У��ɿ����ɼУ�������ˮ�ܽ�����ƿ�е�NH3��ʹƿ��ѹǿ��С����ƿ�����γ�����ѹ�5���ñ�ë����ס��ƿһ��ʱ�䣬���ɿ����ɼС�������������Ҳ�ɣ�
CaCl2+2NH3��+2H2O��2�������ſ�������3��BC��4������ͷ�ι��е�ˮ������ƿ�У��ɿ����ɼУ�������ˮ�ܽ�����ƿ�е�NH3��ʹƿ��ѹǿ��С����ƿ�����γ�����ѹ�5���ñ�ë����ס��ƿһ��ʱ�䣬���ɿ����ɼС�������������Ҳ�ɣ������������1��ʵ�����ü����Ȼ�������ʯ�ҵĹ���������ȡ����������ʽΪ2NH4Cl+Ca��OH��2
 CaCl2+2NH3��+2H2O��2���������ܶȱȿ���С����������ˮ��ֻ���������������ռ����ʴ�Ϊ�������ſ�������3��Ũ����������ԣ�����������Ϊ��������������Ǽ������壬���ԣ�Ũ������������������ܸ��ﰱ�����ʴ�Ϊ��BC��4��������һ�ּ�������ˮ�����壬����ͷ�ι��е�ˮ������ƿ�У�ƿ��ѹǿ��С���������ѹ�������£�ˮ��ѹ��ƿ�ڣ��γ���Ȫ���ʴ�Ϊ������ͷ�ι��е�ˮ������ƿ�У��ɿ����ɼУ�������ˮ�ܽ�����ƿ�е�NH3��ʹƿ��ѹǿ��С����ƿ�����γ�����ѹ�5��Ҫ�γ���Ȫ������ձ��е�Һ��ѹ����ƿ������������������ʹ��ƿ�е�ѹǿ���ʴ�Ϊ���ñ���ë����ס��ƿһ��ʱ�䣬���ɿ����ɼС�
CaCl2+2NH3��+2H2O��2���������ܶȱȿ���С����������ˮ��ֻ���������������ռ����ʴ�Ϊ�������ſ�������3��Ũ����������ԣ�����������Ϊ��������������Ǽ������壬���ԣ�Ũ������������������ܸ��ﰱ�����ʴ�Ϊ��BC��4��������һ�ּ�������ˮ�����壬����ͷ�ι��е�ˮ������ƿ�У�ƿ��ѹǿ��С���������ѹ�������£�ˮ��ѹ��ƿ�ڣ��γ���Ȫ���ʴ�Ϊ������ͷ�ι��е�ˮ������ƿ�У��ɿ����ɼУ�������ˮ�ܽ�����ƿ�е�NH3��ʹƿ��ѹǿ��С����ƿ�����γ�����ѹ�5��Ҫ�γ���Ȫ������ձ��е�Һ��ѹ����ƿ������������������ʹ��ƿ�е�ѹǿ���ʴ�Ϊ���ñ���ë����ס��ƿһ��ʱ�䣬���ɿ����ɼС�
��ϰ��ϵ�д�
�����Ŀ
 KCl��2NH3��
KCl��2NH3��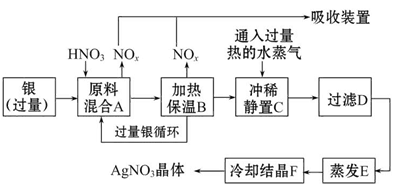
 ��4��������b�Ma��ȡֵ��Χ����֮��Ӧ����Һ�����ʼ������ʵ���������������±��У�
��4��������b�Ma��ȡֵ��Χ����֮��Ӧ����Һ�����ʼ������ʵ���������������±��У�