��Ŀ����
��10�֣���ʡ�Թ�ʢ��ʳ�Σ�������ʳ�ι�ϵ���У�ʳ�����ϰ���������ִ����Ĺ�ũҵ�����о�����Ҫ���ã�û��ʳ�ε������Dz���������ġ������к���Ca2����Mg2����SO42���Լ���ɳ�����ʣ�
��1��Ϊ�˳�ȥ�����е���ɳ���ɲ��õ�ʵ�����������������____��____
��2��Ϊ�˳�ȥ���������ʣ���������ʵ�鲽������ᴿ���ټӹ���BaCl2��Һ���ڼӹ���NaOH��Һ���ۼӹ���Na2CO3��Һ���ܹ��ˣ��ݼ��������ᡣ
���У��ڲ�������漰�����ӷ�Ӧ����ʽ��___ �� ; �� ��
(3)����ʳ����������KIO3��Ϊ�˼���ʵ�����Ƿ�KIO3�����������·�Ӧ���У�
KIO3+5KI+3H2SO4=3I2+3K2SO4+3H2O
Ϊ�˴ӷ�Ӧ�����Һ����ȡ�õ�������I2���ɲ��õ�ʵ�����������������___��__ ��
���𰸡�
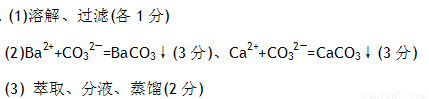
����������

��ϰ��ϵ�д�
�����Ŀ