��Ŀ����
��2013?�㶫����ˮż����Ӧ��һ�����͵�ֱ���������Ӧ�����磺
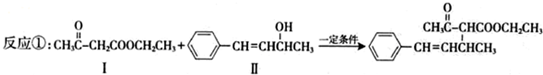
��1���������ķ���ʽΪ
��2����������ʹ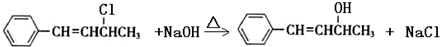
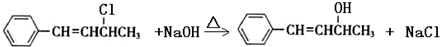 ��
��
��3�����������NaOH�Ҵ���Һ�������ɻ�����������ĺ˴Ź������׳��������������壬�����֮��ΪΪ1��1��1��2�����Ľṹ��ʽΪ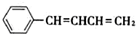
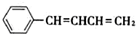 ��
��
��4����CH3COOCH2CH3�ɺϳɻ�������������CH3COOCH2CH3��һ����֧��ͬ���칹�壬̼�����˳ʶԳƽṹ������Cu���������O2��Ӧ�����ܷ���������Ӧ�Ļ�����������Ľṹ��ʽΪ
��5��һ��������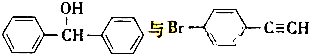 ��Ҳ���Է������Ʒ�Ӧ�ٵķ�Ӧ���л�����Ľṹ��ʽΪ
��Ҳ���Է������Ʒ�Ӧ�ٵķ�Ӧ���л�����Ľṹ��ʽΪ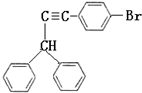
 ��
��

��1���������ķ���ʽΪ
C6H10O3
C6H10O3
��1mol��������ȫȼ��������Ҫ����7
7
molO2����2����������ʹ
��ˮ�����Ը������
��ˮ�����Ը������
��Һ����дһ�֣���ɫ�����������ʽΪC10H11C1������NaOHˮ��Һ�������ɻ��������Ӧ�Ļ�ѧ����ʽΪ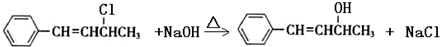
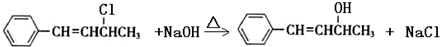
��3�����������NaOH�Ҵ���Һ�������ɻ�����������ĺ˴Ź������׳��������������壬�����֮��ΪΪ1��1��1��2�����Ľṹ��ʽΪ
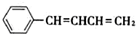
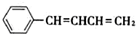
��4����CH3COOCH2CH3�ɺϳɻ�������������CH3COOCH2CH3��һ����֧��ͬ���칹�壬̼�����˳ʶԳƽṹ������Cu���������O2��Ӧ�����ܷ���������Ӧ�Ļ�����������Ľṹ��ʽΪ
HOCH2CH=CHCH2OH
HOCH2CH=CHCH2OH
�����Ľṹ��ʽΪOHCCH=CHCHO
OHCCH=CHCHO
����5��һ��������
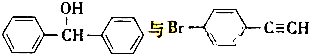 ��Ҳ���Է������Ʒ�Ӧ�ٵķ�Ӧ���л�����Ľṹ��ʽΪ
��Ҳ���Է������Ʒ�Ӧ�ٵķ�Ӧ���л�����Ľṹ��ʽΪ

��������1����Ӧ���л�����I�Ľṹ��ʽΪCH3COCH2COOCH2CH3�����ݽṹ��ʽ�ɵ÷���ʽ��д����Ӧ�ķ���ʽ�ɵú�������
��2����Ӧ���л�����II�Ĺ�������̼̼˫�����ǻ���ǰ��������Է����ӳɷ�Ӧ��ǰ�ߺͺ��߶��ܱ����Ը��������Һ�������ɴ��ƶ�II����ʹ������Ȼ�̼��Һ����ˮ�����Ը��������Һ��ɫ��II�Ľṹ��ʽΪC6H5CH=CHCHOHCH3�������ʽΪC10H12O��III����±�����������ʽΪC10H11Cl��III��II��1��Cl��1��OH��III�� NaOHˮ��Һ��������II��˵��III�Ľṹ��ʽΪC6H5CH=CHCHClCH3������±���������ʿ���д����ƽ��ˮ�ⷴӦ�Ļ�ѧ����ʽ��
��3������±������NaOH�Ҵ���Һ���ȷ�����ȥ��Ӧ��ԭ����III��NaOH�Ҵ���Һ�������ɵ��л����������C6H5CH=C=CHCH3��C6H5CH=CHCH=CH2��ǰ�ߵĺ˴Ź������׳��������������壬�����֮��Ϊ1��1��3����˲��������⣬����IV�Ľṹ��ʽΪC6H5CH=CHCH=CH2��
��4�������Ǽ���-CH2OH��HOCH2-����Cu���������O2��Ӧ�����ܷ���������Ӧ��ȩ����-CHO��OHC-�����������֪V��̼�����˸���1���Ǽ���-CH2OH��HOCH2-����CH3COOCH2CH3�ķ���ʽΪC4H8O2����ȥ2��C��6��H��2��O֮��ɵ�2��C��2��H����V��̼���м�ʣ�����Ϊ-CH=CH-����V�Ľṹ��ʽΪHOCH2CH=CHCH2OH���Դ�Ϊͻ�ƿڣ������ƶ�VI�Ľṹ��ʽΪOHCCH=CHCHO��
��5���ȽϷ�Ӧ���з�Ӧ��I��II��������Ľṹ��ʽ���ҳ�������ͬ�Ļ��źͲ���ͬ�Ļ��ţ�Ȼ���ƶϸ÷�Ӧʵ����II�������ǻ�����̼�����ѣ�I����������ͪ������̼�ϵ�̼������ѣ��ǻ�����������������ˮ�����ಿ�ֽ�������л�����ɴ���ȿɵ����Ʒ�Ӧ�ٵ��л�����Ľṹ��ʽ��
��2����Ӧ���л�����II�Ĺ�������̼̼˫�����ǻ���ǰ��������Է����ӳɷ�Ӧ��ǰ�ߺͺ��߶��ܱ����Ը��������Һ�������ɴ��ƶ�II����ʹ������Ȼ�̼��Һ����ˮ�����Ը��������Һ��ɫ��II�Ľṹ��ʽΪC6H5CH=CHCHOHCH3�������ʽΪC10H12O��III����±�����������ʽΪC10H11Cl��III��II��1��Cl��1��OH��III�� NaOHˮ��Һ��������II��˵��III�Ľṹ��ʽΪC6H5CH=CHCHClCH3������±���������ʿ���д����ƽ��ˮ�ⷴӦ�Ļ�ѧ����ʽ��
��3������±������NaOH�Ҵ���Һ���ȷ�����ȥ��Ӧ��ԭ����III��NaOH�Ҵ���Һ�������ɵ��л����������C6H5CH=C=CHCH3��C6H5CH=CHCH=CH2��ǰ�ߵĺ˴Ź������׳��������������壬�����֮��Ϊ1��1��3����˲��������⣬����IV�Ľṹ��ʽΪC6H5CH=CHCH=CH2��
��4�������Ǽ���-CH2OH��HOCH2-����Cu���������O2��Ӧ�����ܷ���������Ӧ��ȩ����-CHO��OHC-�����������֪V��̼�����˸���1���Ǽ���-CH2OH��HOCH2-����CH3COOCH2CH3�ķ���ʽΪC4H8O2����ȥ2��C��6��H��2��O֮��ɵ�2��C��2��H����V��̼���м�ʣ�����Ϊ-CH=CH-����V�Ľṹ��ʽΪHOCH2CH=CHCH2OH���Դ�Ϊͻ�ƿڣ������ƶ�VI�Ľṹ��ʽΪOHCCH=CHCHO��
��5���ȽϷ�Ӧ���з�Ӧ��I��II��������Ľṹ��ʽ���ҳ�������ͬ�Ļ��źͲ���ͬ�Ļ��ţ�Ȼ���ƶϸ÷�Ӧʵ����II�������ǻ�����̼�����ѣ�I����������ͪ������̼�ϵ�̼������ѣ��ǻ�����������������ˮ�����ಿ�ֽ�������л�����ɴ���ȿɵ����Ʒ�Ӧ�ٵ��л�����Ľṹ��ʽ��
����⣺��1����Ӧ���л�����I�Ľṹ��ʽΪCH3COCH2COOCH2CH3�������ʽΪC6H10O3���������ĺ���������ȼ��ͨʽ�ɵã�C6H10O3+7O2
6CO2+5H2O������������I�����ʵ���֮�ȵ���ϵ��֮�ȣ���1molI��ȫȼ��������Ҫ����7mol O2��
�ʴ�Ϊ��C6H10O3�� 7��
��2��������II�Ĺ�������̼̼˫�����ǻ���ǰ��������Է����ӳɷ�Ӧ��ǰ�ߺͺ��߶��ܱ����Ը��������Һ������III�Ľṹ��ʽΪC6H5CH=CHCHClCH3����NaOH�ڼ��������·���ˮ�ⷴӦ����Ӧ�ķ���ʽΪ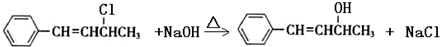 ��
��
�ʴ�Ϊ����ˮ�����Ը�����أ�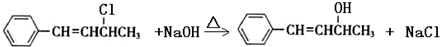 ��
��
��3��III��NaOH�Ҵ���Һ�������ɵ��л����������C6H5CH=C=CHCH3��C6H5CH=CHCH=CH2��ǰ�ߵĺ˴Ź������׳��������������壬�����֮��Ϊ1��1��3����˲��������⣬����IV�Ľṹ��ʽΪ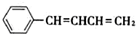 ���ʴ�Ϊ��
���ʴ�Ϊ��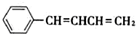 ��
��
��4�������Ǽ���-CH2OH��HOCH2-����Cu���������O2��Ӧ�����ܷ���������Ӧ��ȩ����-CHO��OHC-�����������֪V��̼�����˸���1���Ǽ���-CH2OH��HOCH2-����CH3COOCH2CH3�ķ���ʽΪC4H8O2����ȥ2��C��6��H��2��O֮��ɵ�2��C��2��H����V��̼���м�ʣ�����Ϊ-CH=CH-����V�Ľṹ��ʽΪHOCH2CH=CHCH2OH���Դ�Ϊͻ�ƿڣ������ƶ�VI�Ľṹ��ʽΪOHCCH=CHCHO���ʴ�Ϊ��HOCH2CH=CHCH2OH�� OHCCH=CHCHO��
��5���������Ϣ��֪��Ӧʵ����II�������ǻ�����̼�����ѣ�I����������ͪ������̼�ϵ�̼������ѣ��ǻ�����������������ˮ����������Ľṹ��ʽΪ ��
��
�ʴ�Ϊ�� ��
��
| ��ȼ |
�ʴ�Ϊ��C6H10O3�� 7��
��2��������II�Ĺ�������̼̼˫�����ǻ���ǰ��������Է����ӳɷ�Ӧ��ǰ�ߺͺ��߶��ܱ����Ը��������Һ������III�Ľṹ��ʽΪC6H5CH=CHCHClCH3����NaOH�ڼ��������·���ˮ�ⷴӦ����Ӧ�ķ���ʽΪ
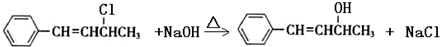 ��
���ʴ�Ϊ����ˮ�����Ը�����أ�
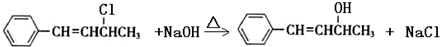 ��
����3��III��NaOH�Ҵ���Һ�������ɵ��л����������C6H5CH=C=CHCH3��C6H5CH=CHCH=CH2��ǰ�ߵĺ˴Ź������׳��������������壬�����֮��Ϊ1��1��3����˲��������⣬����IV�Ľṹ��ʽΪ
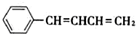 ���ʴ�Ϊ��
���ʴ�Ϊ��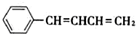 ��
����4�������Ǽ���-CH2OH��HOCH2-����Cu���������O2��Ӧ�����ܷ���������Ӧ��ȩ����-CHO��OHC-�����������֪V��̼�����˸���1���Ǽ���-CH2OH��HOCH2-����CH3COOCH2CH3�ķ���ʽΪC4H8O2����ȥ2��C��6��H��2��O֮��ɵ�2��C��2��H����V��̼���м�ʣ�����Ϊ-CH=CH-����V�Ľṹ��ʽΪHOCH2CH=CHCH2OH���Դ�Ϊͻ�ƿڣ������ƶ�VI�Ľṹ��ʽΪOHCCH=CHCHO���ʴ�Ϊ��HOCH2CH=CHCH2OH�� OHCCH=CHCHO��
��5���������Ϣ��֪��Ӧʵ����II�������ǻ�����̼�����ѣ�I����������ͪ������̼�ϵ�̼������ѣ��ǻ�����������������ˮ����������Ľṹ��ʽΪ
 ��
���ʴ�Ϊ��
 ��
�����������⿼�鿼����ϩ����Ȳ����±����������ȩ�����ȳ����л���Ľṹ�ͳɼ��������˽⣻���鿼���������л�����ʹ��������ɺ������Լ��������ϵ���˽⣻���鿼������Ҫ�л���Ӧ������ȡ����Ӧ���ӳɷ�Ӧ����ȥ��Ӧ��������Ӧ���˽⣻���鿼���Ժ˴Ź������������ͷ����֮�ȵ��˽⣻���鿼�����л�������ͬ���칹������˽⣻���鿼���ķ����������������������ۺ�Ӧ����Ϣ�Լ���ϢǨ��������

��ϰ��ϵ�д�
�����Ŀ