��Ŀ����
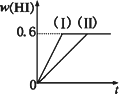
��1 mol��I2(g)��2 mol��H2(g)����ij2 L�ܱ������У���һ���¶��·�����Ӧ��I2(g)��H2(g)![]() 2HI(g)����H��0�����ﵽƽ�⣮HI�����������(HI)��ʱ��仯��ͼ����(��)��ʾ��
2HI(g)����H��0�����ﵽƽ�⣮HI�����������(HI)��ʱ��仯��ͼ����(��)��ʾ��
(1)��ƽ��ʱ��I2(g)�����ʵ���Ũ��Ϊ________mol��L��1��
(2)���ı䷴Ӧ��������ij��������(HI)�ı仯������(��)��ʾ���������������________(�����������������)��
�ٺ��������£������¶�
�ں��������£������¶�
�ۺ��������£���С��Ӧ�������
�ܺ��������£�����Ӧ�������
�ݺ��¡����������£������ʵ�����
(3)�������¶Ȳ��䣬����һ��ͬ��2 L�ܱ������м���a mol��I2(g)��b mol��H2(g)��c mol��HI(g)(a��b��c������0)��������Ӧ��ƽ��ʱ��HI�����������Ϊ0.60����a��b��c�Ĺ�ϵΪ________(��һ����a��b��c�Ĵ���ʽ��ʾ)��
������
����(1)0.05��
����(2)�ۢݣ�
����(3)2b��4a��c

|
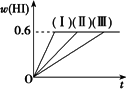
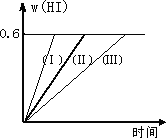
һ���¶��£���1 mol��I2(g)��2 mol��H2����2 L�ܱ������У�������Ӧ��I2(g)��H2(g) �ٺ��������£������¶� �ں��������£���С��Ӧ������� �ۺ��º��������£������ʵ����� �ܺ��º��������£��ټ���1 mol��I2(g)��2 mol��H2 �ݺ��������£�����Ӧ�������
| |
| [����] | |
A�� |
�٢� |
B�� |
�٢ڢ� |
C�� |
�ڢۢ� |
D�� |
�ۢ� |
 2HI(g)����H��0�����ﵽƽ�⣮HI���������w(HI)��ʱ��仯������(��)��ʾ���ı�ijһ����������w(HI)�仯������(��)��ʾ���ı�������ǣ�
2HI(g)����H��0�����ﵽƽ�⣮HI���������w(HI)��ʱ��仯������(��)��ʾ���ı�ijһ����������w(HI)�仯������(��)��ʾ���ı�������ǣ�