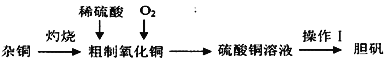
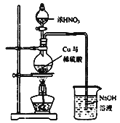
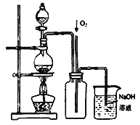
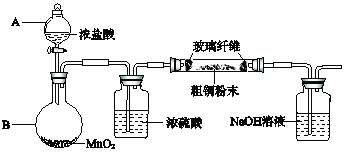
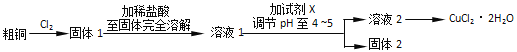��Ŀ����
����Ŀ��������CH3OCH3�ֳƼ��ѣ��۵�-141.5�棬�е�-24.9�档�ɺϳ���(CO��H2)�Ʊ������ѵķ�Ӧԭ�����£�
��CO(g)+2H2(g) ![]() CH3OH(g) ��H1 =-90.0 kJ��mol-1
CH3OH(g) ��H1 =-90.0 kJ��mol-1
��2CH3OH(g) ![]() CH3OCH3(g)+ H2O(g) ��H2 = -20.0 kJ��mol-1
CH3OCH3(g)+ H2O(g) ��H2 = -20.0 kJ��mol-1
�ش��������⣺
(1)��Ӧ����_____________(����¡����¡�)�����Է����С�
(2)д���ɺϳ���(CO��H2)ֱ���Ʊ�CH3OCH3���Ȼ�ѧ����ʽ��______________��
(3)�¶�Ϊ500Kʱ����2L���ܱ������г���2mol CO��6molH2������Ӧ�١��ڣ�5minʱ�ﵽƽ�⣬ƽ��ʱCO��ת����Ϊ60%��c(CH3OCH3) = 0.2 mol��L-1����H2��ʾ��Ӧ�ٵ�������________����Ӧ�ڵ�ƽ�ⳣ��K =____________��
(4)�о����֣��������ͬ�������м������ʵ�����ͬ��CO��H2������Ӧ�١��ڣ��ڲ�ͬ�¶Ⱥ�����������¾�����ͬ��Ӧʱ��������ʵ�����ݣ�
T(K) | ���� | COת��(��) | CH3OCH3ѡ����(��) |
473 | �� | 10 | 36 |
500 | �� | 12 | 39 |
500 | Cu/ZnO | 20 | 81 |
����ע��������ѡ���ԣ�ת����CO������CH3OCH3�ٷֱ�
����ͬ�¶��£�ѡ��Cu/ZnO���������ô�����_______ (����)��
A���ٽ�ƽ�������ƶ� B����߷�Ӧ���� C�����ͷ�Ӧ�Ļ��
D���ı䷴Ӧ���ʱ� E.���CO��ƽ��ת����
�ڱ���ʵ�����ݱ�������500Kʱ������Cu/ZnO��COת����CH3OCH3��ѡ������������Ӱ�죬��ԭ����__________________________��
���𰸡����� 2CO(g)+4H2(g)= CH3OCH3(g) + H2O(g) ��H =-200 kJ��mol-1 0.24mol/(L��min) 1 B��C �ɱ������ݿ�֪����ʱ��Ӧ��ϵ��δ����ƽ�⣬�����ɼ��ٷ�Ӧ�ڣ��Ӷ�����ͬʱ�������ɸ����CH3OCH3
��������
���⿼����ǻ�ѧƽ��֪ʶ��
��1����Ӧ���Ƿ��ȵ������С�Ŀ��淴Ӧ���������G=��H-T����S��֪�ڵ��������Է����С�
��2�����ݸ�˹���ɿ�֪����2+�ڼ��õ��ɺϳ�����CO��H2��ֱ���Ʊ�CH3OCH3���Ȼ�ѧ��Ӧ����ʽΪ2CO(g)+4H2(g)= CH3OCH3(g) + H2O(g)��H=-200 kJ��mol-1��
��3����ƽ�������ʽ�������H2��ʾ��Ӧ�ٵ����ʺͿ��淴Ӧ�ڵ�ƽ�ⳣ��K��
��1����Ӧ���Ƿ��ȵ������С�Ŀ��淴Ӧ���������G=��H-T����S��֪�ڵ��������Է����С���С���Ϊ�����¡�
��2�����ݸ�˹���ɿ�֪����2+�ڼ��õ��ɺϳ�����CO��H2��ֱ���Ʊ�CH3OCH3���Ȼ�ѧ��Ӧ����ʽΪ2CO(g)+4H2(g)= CH3OCH3(g) + H2O(g)��H=-200 kJ��mol-1����С���Ϊ��2CO(g)+4H2(g)= CH3OCH3(g) + H2O(g)��H=-200 kJ��mol-1��
��3���¶�Ϊ500Kʱ����2L���ܱ������г���2mol CO��6molH2������Ӧ��������5minʱ�ﵽƽ�⣬ƽ��ʱCO��ת����Ϊ60%��������CO��1.2mol����������������2.4mol��Ũ����1.2mol/L����H2��ʾ��Ӧ�ٵ�������1.2mol/L��5min=0.24mol/(L��min)��ƽ��ʱc(CH3OCH3) = 0.2 mol��L-1������ˮ������Ũ��Ҳ��0.2 mol��L-1���״���Ũ����0.6mol/L-0.2mol/L��2=0.2mol/L�����Է�Ӧ�ڵ�ƽ�ⳣ��K=��0.2��0.2��/0.22=1����С���Ϊ��0.24mol/(L��min)��1��
��4����A.�������ܸı�ƽ��״̬����A����
B.����������߷�Ӧ���ʣ���B��ȷ��
C.�������Խ��ͷ�Ӧ�Ļ�ܣ���C��ȷ��
D.�������ܸı䷴Ӧ���ʱ䣬��D����
E.�������ܸı�ƽ��״̬�����������CO��ƽ��ת���ʣ���E����ѡB��C��
���ɱ������ݿ�֪����ʱ��Ӧ��ϵ��δ����ƽ�⣬�����ɼ��ٷ�Ӧ�ڣ��Ӷ�����ͬʱ�������ɸ����CH3OCH3����С���Ϊ���ɱ������ݿ�֪����ʱ��Ӧ��ϵ��δ����ƽ�⣬�����ɼ��ٷ�Ӧ�ڣ��Ӷ�����ͬʱ�������ɸ����CH3OCH3��

 �·Ƿ��̸����100��ϵ�д�
�·Ƿ��̸����100��ϵ�д�����Ŀ����֪ij���巴Ӧ��ƽ�ⳣ���ɱ�ʾΪK=c(CH3OCH3)c(H2O)/c2(CH3OH)���÷�Ӧ�ڲ�ͬ�¶��µ�ƽ�ⳣ����400����K=32��500����K=44����ش��������⣺
��1��д��������Ӧ�Ļ�ѧ����ʽ��_________________________________ ��
��2���÷�Ӧ����H _________0��
��3����֪���ܱ������У����ijʱ�̸���ֵ�Ũ�����£�
���� | CH3OH(g�� | CH3OCH3(g�� | H2O(g�� |
Ũ��/��molL-1�� | 0.54 | 0.68 | 0.68 |
�ٴ�ʱ�¶�400�棬��ijʱ������_______���������������ͬ����
�������¶�Ϊ�����꣬�Ը��¶���ƽ��̬�״����ʵ���nΪ�����꣬��ʱ��Ӧ����ͼ���λ����ͼ��____�㣬�Ƚ�ͼ��B��D��������Ӧ������Ӧ������B_______��D��������____��

��4��һ��������Ҫ��߷�Ӧ���ת���ʣ����Բ��õĴ�ʩ��___________��
a�������¶� b��������� c��ѹ�����������
d������ˮ������Ũ�� e����ʱ���������