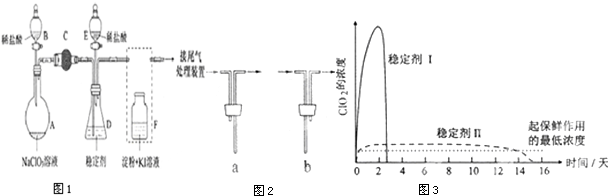
��Ŀ����
5����һƿ��ɫ������Һ�����ܺ�NH4+��K+��Na +��Mg2+��Ba2+��Al3+��Fe3+��SO42-��CO32-��Cl-��I-�е�һ�ֻ��֣�ȡ����Һ��������ʵ�飺
����pH��ֽ���飬������Һ�����ԣ�
��ȡ������Һ����������CCl4���������Ƶ���ˮ�������ã�CCl4����Ϻ�ɫ��
��ȡ������Һ����μ���ϡNaOH��Һ��ʹ��Һ��������ת��Ϊ���ԣ��ڵμӹ����м��μ���Ϻ���Һ�о��������ɣ�
��ȡ���������ʼ��Ե���Һ����Na2CO3��Һ���а�ɫ�������ɣ�
�ݽ��۵õ��ļ�����Һ���ȣ�������ų�����������ʹʪ��ĺ�ɫʯ����ֽ������
��������ʵ����ʵȷ�����ش�����Һ�п϶������ڵ�������SO42-��CO32-��Mg2+��Al3+��Fe3+��Ҫȷ���Ƿ���ڵ������ӵ�ʵ�鷽����ȡ������Һ���Թ��У��μ�ϡ���ᣬ�ٵμ���������Һ������а�ɫ�������֣�˵�����и������ӣ������и������ӣ�
���� ��ɫ��Һ�в������Fe3+������pH��ֽ������Һ�����ԣ�������Һ�в�������ǿ�ᷴӦ��CO32-���ӣ��ڵ������ܺ���ˮ��Ӧ���ɵⵥ�ʣ��������Ȼ�̼�г���ɫ��ͨ��ʵ�������жϣ���Һ�к���I-����þ���ӡ������ӡ������Ӷ����������Ʒ�Ӧ���ɳ�����ͨ��ʵ�������жϣ���Һ�в����������������ӷ�Ӧ���ɳ�����Mg2+��Al3+��Fe3+���ܱ����Ӻ�̼���Ʒ�Ӧ���ɰ�ɫ������ͨ��ʵ�������жϣ���Һ������Na2CO3��Һ��Ӧ���ɰ�ɫ������Ba2+�������Ba2+���ӣ���Һ�о�û��SO42-����笠������ڼ������������������Ʒ�Ӧ������ʹʪ��ĺ�ɫʯ����ֽ����ɫ�İ���������笠����ӵ�������Ӧ��ͨ��ʵ�������жϣ���Һ����NH4+���ݴ˽��н��
��� �⣺��ɫ��Һ�в������Fe3+��
����Һ��ǿ���ԣ�������Һ�в�������ǿ�ᷴӦ��CO32-���ӣ�
�ڵ������ܺ���ˮ��Ӧ���ɵⵥ�ʣ��������Ȼ�̼�г���ɫ��ͨ��ʵ�������жϣ���Һ�к���I-��
��þ���ӡ������ӡ������Ӷ����������Ʒ�Ӧ���ɳ�����ͨ��ʵ�������жϣ���Һ�в����������������ӷ�Ӧ���ɳ�����Mg2+��Al3+��Fe3+��
�ܱ����Ӻ�̼���Ʒ�Ӧ���ɰ�ɫ������ͨ��ʵ�������жϣ���Һ������Na2CO3��Һ��Ӧ���ɰ�ɫ������Ba2+�������Ba2+���ӣ���Һ�о�û��SO42-��
��笠������ڼ������������������Ʒ�Ӧ������ʹʪ��ĺ�ɫʯ����ֽ����ɫ�İ���������笠����ӵ�������Ӧ��ͨ��ʵ�������жϣ���Һ����NH4+��
ͨ�������������ж���Һ���Ƿ���K+��Na+��Cl-��
��������Һ�п϶����ڵ�������I-��NH4+��Ba2+���϶������ڵ�������SO42-��CO32-��Mg2+��Al3+��Fe3+��������ȷ���Ƿ���ڵ�������K+��Na+��Cl-��
ȷ�������������ӵĴ��ڵķ�����ȡ������Һ���Թ��У��μ�ϡ���ᣬ�ٵμ���������Һ������а�ɫ�������֣�˵�����и������ӣ������и������ӣ�
�ʴ�Ϊ��SO42-��CO32-��Mg2+��Al3+��Fe3+��ȡ������Һ���Թ��У��μ�ϡ���ᣬ�ٵμ���������Һ������а�ɫ�������֣�˵�����и������ӣ������и������ӣ�
���� ���⿼���˳������ӵ����ʼ����鷽������Ŀ�Ѷ��еȣ���ȷ�������ӵ�����Ϊ���ؼ���ע���������ճ����ڵļ��鷽��������������ѧ���ķ���������������������������

| A�� | 0.1mo/L | B�� | 0.15mol/L | C�� | 0.2mol/L | D�� | 0.3mol/L |
 2014�����籭�������ڰ������У�2016������ֽ��ٰ�31����˻ᣮ�����廷������ȫ��������������Ž���һ�����и����е����ʣ���������ͼ����Ӱ���ֹ�ϵ���ǣ�������
2014�����籭�������ڰ������У�2016������ֽ��ٰ�31����˻ᣮ�����廷������ȫ��������������Ž���һ�����и����е����ʣ���������ͼ����Ӱ���ֹ�ϵ���ǣ�������| �� | �� | �� | �� | |
| A | NaCl | K2SO4 | KCl | ��NH4��2SO4 |
| B | Na2SO4 | K2SO4 | KCl | NH4Cl |
| C | NaCl | K2SO4 | KCl | NH4Cl |
| D | Na2SO4 | K2SO4 | KCl | ��NH4��2SO4 |
| A�� | A�� | B�� | B�� | C�� | C�� | D�� | D�� |
| A�� | 0��l mol•L-1��NaAlO2��Һ��K+��Na+��SO42-��CO32- | |
| B�� | ʹpH��ֽ��������Һ�У�NH4+��Na+��SO42-��Cl- | |
| C�� | ���д���ClO- ��Һ�У�K+��OH-��I-��SO32- | |
| D�� | c��Al3+��=0.1 mol•L-1����Һ�У�Na+��Cl-��AlO2-��SO42- |
| A�� | 0.5 mol | B�� | 1 mol | C�� | 1.5 mol | D�� | 2 mol |
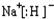 ��CA��ˮ��Ӧ�ų�����Ļ�ѧ����ʽΪNaH+H2O=NaOH+H2����
��CA��ˮ��Ӧ�ų�����Ļ�ѧ����ʽΪNaH+H2O=NaOH+H2����