��Ŀ����
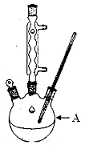
�屽��һ�ֻ���ԭ�ϣ�ʵ���Һϳ��屽��װ��ʾ��ͼ���й��������£�

�����кϳɲ���ش����⣺
(1)��a�м���15 mL��ˮ����������м����b��С�ļ���4.0 mLҺ̬�塣��a�е��뼸���壬�а�ɫ��������������Ϊ������ ���塣�����μ���Һ����ꡣװ��d�������� ��
(2)Һ�����������в�������ᴿ��
����a�м���10 mLˮ��Ȼ����˳�ȥδ��Ӧ����м��
����Һ������10 mLˮ��8 mL 10%��NaOH��Һ��10 mLˮϴ�ӡ�NaOH��Һϴ�ӵ������� ��
����ֳ��Ĵ��屽�м�����������ˮ�Ȼ��ƣ����á����ˡ������Ȼ��Ƶ�Ŀ���� ��
(3)�����Ϸ���������屽�л����е���Ҫ����Ϊ ��Ҫ��һ���ᴿ�����в����б������ (������ȷѡ��ǰ����ĸ)��
A���ؽᾧ B������ C������ D����ȡ
(4)�ڸ�ʵ���У�a���ݻ����ʺϵ��� (������ȷѡ��ǰ����ĸ)��
A��25 mL B��50 mL C��250 mL D��500 mL

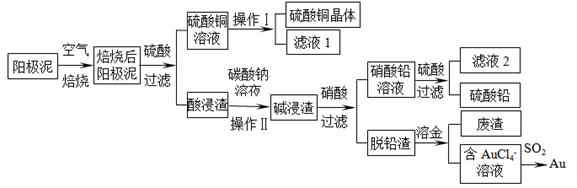
| | �� | �� | �屽 |
| �ܶ�/(g��cm��3) | 0.88 | 3.10 | 1.50 |
| �е�/�� | 80 | 59 | 156 |
| ˮ���ܽ�� | �� | �� | �� |
�����кϳɲ���ش����⣺
(1)��a�м���15 mL��ˮ����������м����b��С�ļ���4.0 mLҺ̬�塣��a�е��뼸���壬�а�ɫ��������������Ϊ������ ���塣�����μ���Һ����ꡣװ��d�������� ��
(2)Һ�����������в�������ᴿ��
����a�м���10 mLˮ��Ȼ����˳�ȥδ��Ӧ����м��
����Һ������10 mLˮ��8 mL 10%��NaOH��Һ��10 mLˮϴ�ӡ�NaOH��Һϴ�ӵ������� ��
����ֳ��Ĵ��屽�м�����������ˮ�Ȼ��ƣ����á����ˡ������Ȼ��Ƶ�Ŀ���� ��
(3)�����Ϸ���������屽�л����е���Ҫ����Ϊ ��Ҫ��һ���ᴿ�����в����б������ (������ȷѡ��ǰ����ĸ)��
A���ؽᾧ B������ C������ D����ȡ
(4)�ڸ�ʵ���У�a���ݻ����ʺϵ��� (������ȷѡ��ǰ����ĸ)��
A��25 mL B��50 mL C��250 mL D��500 mL
(1)HBr������HBr��Br2��(2)�ڳ�ȥHBr��δ��Ӧ��Br2���۸���
(3)����C��(4)B
(3)����C��(4)B
���ݱ����巴Ӧ�Ʊ��屽������֪��м������ã�Ȼ��Բ�����з�����ᴿ��
(1)�������ڴ��������·�Ӧ��������HBr���лӷ��ԣ������а������������ڸ÷�Ӧ���ȣ���Һ����лӷ��ԣ��ʻӷ�����������Br2��HBr����NaOH��Һ���ա�
(2)(3)��Ӧ��Ϻ�Ӧ�����е���Ҫ�ɷ����屽������δ��Ӧ�ı���Br2�����ɵ�HBr������NaOH��ȥHBr��δ��Ӧ��Br2����ˮϴ����CaCl2��һ�����и�������ݱ����屽�ķе㲻ͬ����������롣
(4)��ƿ��Ϊ��Ӧ����ʱ��Һ�������������ݻ���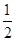 ����Һ������Ϊ15 mL��4 mL��19 mL�����ݻ�ѡ��50 mL��
����Һ������Ϊ15 mL��4 mL��19 mL�����ݻ�ѡ��50 mL��
(1)�������ڴ��������·�Ӧ��������HBr���лӷ��ԣ������а������������ڸ÷�Ӧ���ȣ���Һ����лӷ��ԣ��ʻӷ�����������Br2��HBr����NaOH��Һ���ա�
(2)(3)��Ӧ��Ϻ�Ӧ�����е���Ҫ�ɷ����屽������δ��Ӧ�ı���Br2�����ɵ�HBr������NaOH��ȥHBr��δ��Ӧ��Br2����ˮϴ����CaCl2��һ�����и�������ݱ����屽�ķе㲻ͬ����������롣
(4)��ƿ��Ϊ��Ӧ����ʱ��Һ�������������ݻ���
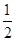 ����Һ������Ϊ15 mL��4 mL��19 mL�����ݻ�ѡ��50 mL��
����Һ������Ϊ15 mL��4 mL��19 mL�����ݻ�ѡ��50 mL��
��ϰ��ϵ�д�
�����Ŀ